Chlorine dioxide solutions for cooling tower water treatment sector - Water and Wastewater - Water Treatment
Chlorine dioxide provides a safe and cost-effective approach to cooling tower water treatment. While there are other oxidation chemistries available to use in heavy industrial water treatment (e.g. bleach, bromine or chlorine), none offer the combination of performance, safety, shelf-life, environmental and cost benefits like ClO2.
Better Microbiological Control
It is not an exaggeration to say that at least 80% of all cooling water problems relate to poor microbiological control, especially for hydrocarbon processing industries, like refining, petrochemical and ammonia. Microbial growth results in serious problems for these industries, including loss of heat transfer in heat exchangers, a collapse of cooling tower fill, equipment corrosion and more.
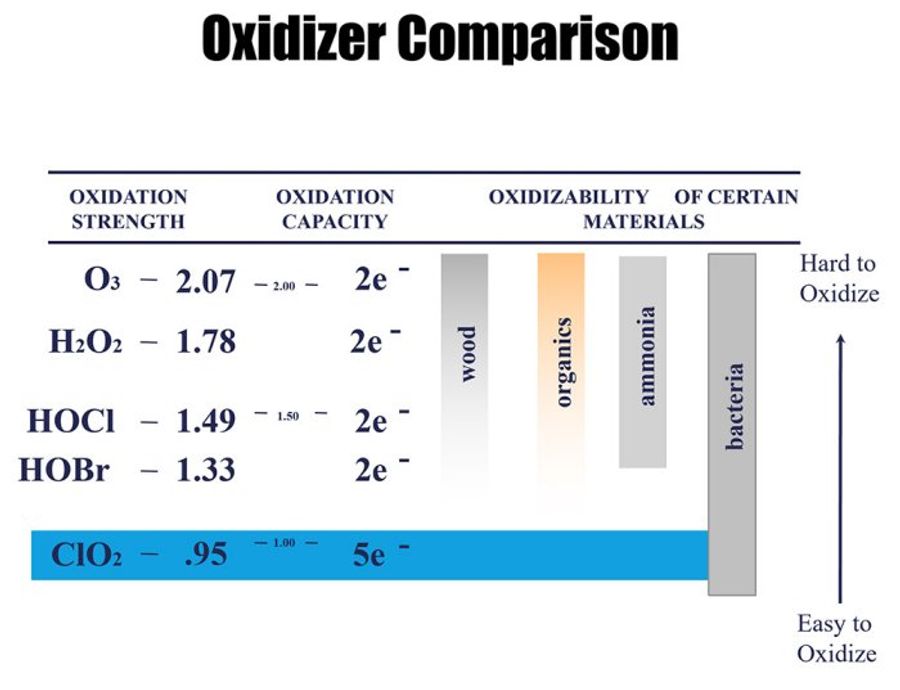
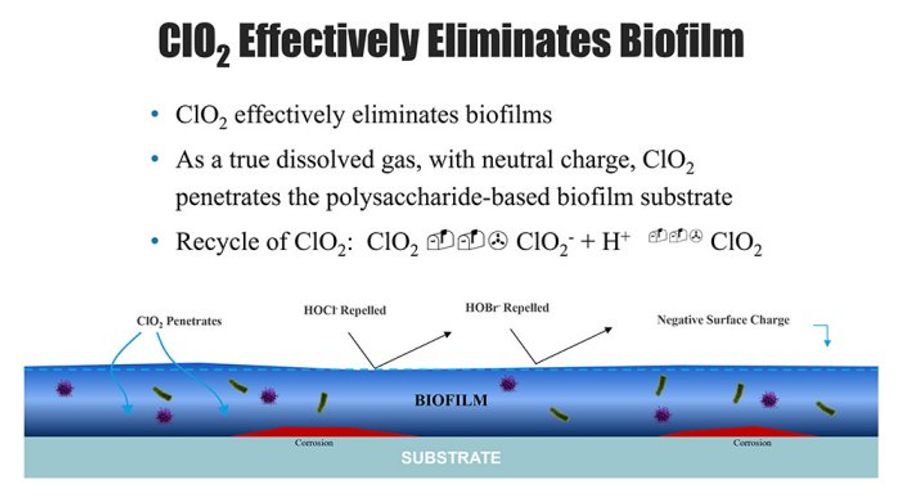
Chlorine dioxide is an ideal solution for microbiological control because it:
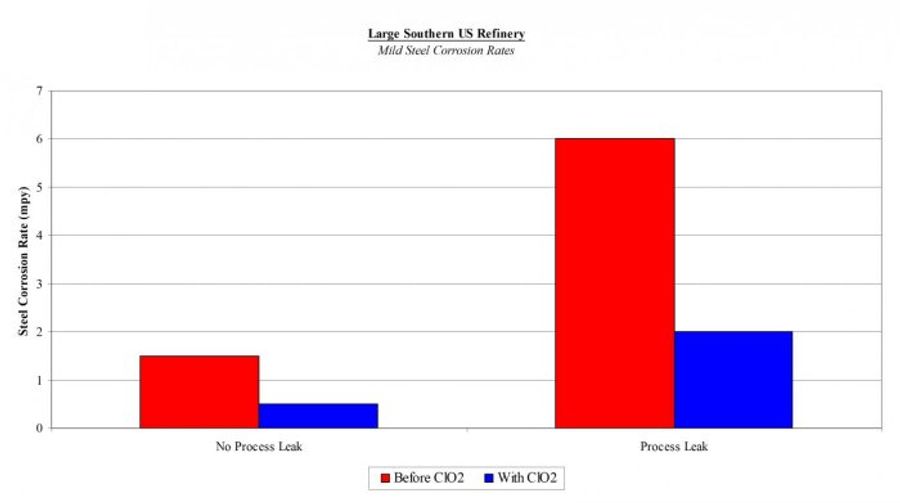
Is up to 50% less corrosive than other oxidants
For a relative cost comparison, if the feed rate of bleach (as chlorine) is at or above an average of 0.2 ppm based on the recirculation rate in the cooling tower, ClO2 can be fed competitively in this range under normal circumstances.
Effective Emulsion Breaking
Most oil/water emulsions are stabilized by FeS and/or biomass. Chlorine dioxide efficiently eliminates both, thus destabilizing the emulsion and breaking it up.
H2S
Chlorine dioxide destroys H2S by oxidizing it to sulfate. This permanently eliminates the compound, rather than simply adsorbing it into amine as most other H2S treatment chemistries do. These amine based chemistries will also release the H2S again if the pH varies from its operating range.
In addition, most H2S is generated from sulfate reducing bacteria (SRB’s). ClO2 is especially effective at killing these bacteria and eliminating the source of additional H2S.
Phenol Destruction
Chlorine dioxide is very effective at the destruction of phenol, converting it to benzoquinone and hydroquinone, which are environmentally benign. The reaction is fast, but pH dependent. Dosages range from 1.5 ppm ClO2/ppm phenol at pH less than 10.0 to 3.3 ppm ClO2/ppm phenol for pH greater than 10.0.
Cost-Effective Economics
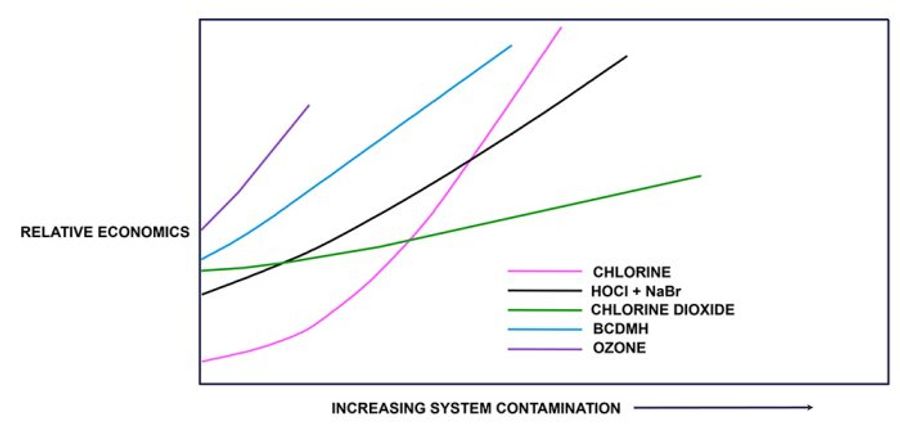
Chlorine dioxide’s selective reactivity results in favorable cost-effective economics compared to alternative oxidizing biocides, especially when the system is heavily contaminated.
Cooling Tower Water Treatment Supplies and Expertise
International Dioxcide is the leading producer of chlorine dioxide precursor chemistries and ClO2 generation systems in North America. We provide safe, reliable, and cost-effective water treatment solutions for commercial applications. Our skilled team of engineers, field technicians, and customer service personnel design, build, and implement custom ClO2 solutions for each treatment site.