Invitro Toxicity Monitoring Services For Monitoring And Testing Available In Bhutan
-
Manufactured by ImmunoPrecise Antibodies Ltd.based in CANADA
In vitro analytical tools allow for the study of various critical quality attributes like production yield, target- and/or poly-reactivity, colloidal stability, melting temperature, self- and/or cross-interaction, solubility, and fragmentation. IPA is equipped with a state-of-the-art bio-inert HPLC enabling analytical determination of mAb biochemical and biophysical properties ...
-
Manufactured by Calbiotech Inc.based in
Calbiotech offers contract manufacturing of cGMP compliant, fully traceable, supply-chain-managed buffers and reagents, providing an economical outsourced alternative from a dependable and stable source. We have been designing, developing and manufacturing custom diagnostic reagents and kits since 1998. We produce in vitro diagnostic controls and reagents, custom processed human and animal serums ...
-
Manufactured by Intracare BVbased in NETHERLANDS
Quality and research are key factors at Intracare; In our state-of-the-art laboratory a solid quality control testing program is performed. In addition, research analysis are conducted. The laboratory includes a physical chemical laboratory and a microbiological laboratory. All crucial product parameters are analyzed with modern techniques e.g.: HPLC, AES and PCR. At our laboratory we have five ...
-
based in BELGIUM
High capacity and short turn around time: The chemical analysis service comprises raw materials, APIs, finished products, packaging material and medical devices. Eurofins has a network of experienced chemists, well-equipped laboratories and well-structured processes allowing us to offer an exceptional short turnaround time. For example, Eurofins' pharma services operate more than 15 GC and 40 ...
-
Manufactured by Piramal Pharma Solutions (PPS)based in INDIA
In vitro facilities at PDS provide absorption, distribution, metabolism, and excretion (ADME) services for faster and improved decision making points or standalone offering to support drug discovery programs. Cost-efficient, standardized and customized assays for ADME parameters are provided to the ...
-
Manufactured by AventaCell BioMedical Corpbased in USA
Mesenchymal stem cells (MSC) are multipotent cells with regenerative, multidifferentiation, and immunomodulatory capacities. Expanding MSC for clinical purposes usually requires large cell dose, which demanding efficient cultivation ...
-
Manufactured by Regis Technologies Inc.based in USA
HPLC Tools for Drug Membrane Permeability Prediction. IAM Drug Discovery HPLC Column is an ideal tool for high throughput prediction of drug membrane permeability. The results from this column correlate well to traditional in-vitro methods such as intestinal tissue and Caco-2 Cells, yet are faster and cheaper to achieve. Phosphatidylcholine (PC) is the malor phospholipid found in cell membranes. ...
-
Manufactured by Piramal Pharma Solutions (PPS)based in INDIA
Piramal’s in vitro Biology is designed to provide best in class support to our clients to enable their preclinical drug discovery, including assay development, hit identification, hit-to-lead and lead optimization, including HTS, multiplex assay development, primary/secondary functional assays, HCS, and selectivity. Our assay team can develop, and screen compounds, with miniaturization of ...
-
Manufactured by AnaBios Corporationbased in USA
Measuring Neurotoxicity in Compounds Using Peripheral Sensory Neurons. The neurotoxicity of compounds on peripheral sensory neurons can be evaluated with human dorsal root ganglia (DRG) cultures at AnaBios. Clinical use of chemotherapeutic agents can be limited by the development of painful sensory neuropathy. Dissociated human DRG neuronal culture can be used as an in-vitro model of ...
-
Manufactured by AB Sciex Pte. Ltdbased in USA
Simplify Service and Maintence of Your Clinical Mass Spec System. All SCIEX Medical Device Service Professionals follow a 4-step training program and are certified (and re-certified every 2 years) on SCIEX in vitro diagnostic instruments as well as all major HPLC ...
-
Manufactured by Creative Bioarraybased in USA
Immortalized cells are a population of cells from a multicellular organism due to mutation, which can escape normal cellular senescence and keep undergoing division. Thus, this kind of cells can grow in vitro for prolonged periods. The mutations required for immortality can occur naturally or be intentionally induced for experimental purposes. Immortal cell lines are a powerful tool for the ...
-
Manufactured by Creative Bioarraybased in USA
In vitro toxicology and in vitro toxicity testing involves the study and interpretation of adverse effects through in vitro research methods typically associated with bacterial and mammalian cell cultures. Creative Bioarray's in vitro toxicity testing uses the latest advances in the field of in vitro toxicology to help clients determine the viability of compounds early in the preclinical phase ...
-
Manufactured by Gentronix Ltdbased in UNITED KINGDOM
The in vitro micronucleus test (MNT) is a main-stay of most regulatory genotoxicity testing strategies, and is used to detect in vitro clastogens and aneugens. In this regard it complements the Ames test well, as between the two study types they detect all of the three major classes of genotoxin. Fluorescence In Situ Hybridisation (FISH) can also be conducted to differentiate predominantly ...
-
Manufactured by Molecular Biology Resources, Inc.based in USA
MBR offers assay services to investigate the effects of drug compounds on key DNA replication and repair enzymes, such as human DNA polymerase α (alpha), human DNA polymerase β (beta), human DNA polymerase γ (gamma), and HeLa cell extracts (containing replication protein ...
-
based in GERMANY
For legal manufacturers of in vitro diagnostic devices we offer conformity assessment procedures according to Directive 98/79/EC as a Notified Body (EU Identification Number „0483“).Conformity assessment with the involvement of a Notified Body is mandatory for all manufacturers of products according to Annex II list A and B, as well as, for self testing products by lay ...
-
based in UNITED KINGDOM
It is inevitable that there will be occasions where the existing dataset is not sufficient to allow for a robust hazard characterisation and risk assessment. At other times, a request from a regulator may leave no alternative but to commission additional work. Whatever the context, we can offer advice on a testing programme, from protocol to the final report. ...
-
based in UNITED KINGDOM
No matter what business or industry you’re in,ShPhas a solution to help you manage and comply with ...
-
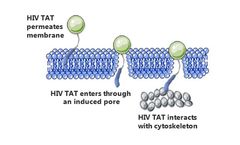
Manufactured by Creative Peptidesbased in USA
Cell penetrating peptides (CPPs), also known as protein transduction domains (PTDs) or membrane transduction peptides (MTPs), are small molecular peptides that consist of 5-30 amino acid residues, which not only penetrate the cell membrane by themselves, but also carry a variety of exogenous substances such as peptides, proteins, nucleic acids and nanoparticles in a covalent or non-covalent ...
-
Manufactured by Creative Peptidesbased in USA
Peptides have inherent properties such as high biological activity, low toxicity, and high specificity. Peptides are now increasingly used in clinical research. A number of peptide drugs are already on the market, and hundreds more are at different stages of clinical development. And GMP peptides can help accelerate the peptide drug development process in the early stages of drug ...
-
based in UNITED KINGDOM
Adventitious agent and mycoplasma testing services from SGS – biologics safety testing to ensure that your cell cultures, virus stocks and final products are certified free from contaminating agents, such as endotoxins and viruses. During the development of your biopharmaceutical products, the threat of cell culture contamination by adventitious agents such as mycoplasma, endotoxins and ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you