nZVI Tester - Device for Measurement of nZVI Content in a Slurry
From Equipment
Simple device for measurement of reactive zero-valent iron content in a slurry or powder.
-
Most popular related searches
Product Details
nZVI TESTER provides simple and fast measurement of zero-valent iron concentration in a product, even under the field conditions. The apparatus can be used for analysis of nZVI content in dry powder or slurry.
The method is based on the measurement of hydrogen volume, which is evolving during chemical reaction ofzero-valent iron and an acid. This test is very simple and particularly fair minded: the volume of hydrogen is directly proportional to the amount of ZVI. The amount of iron-oxides does not influence the volume of generated hydrogen, it only slows down the reaction speed.
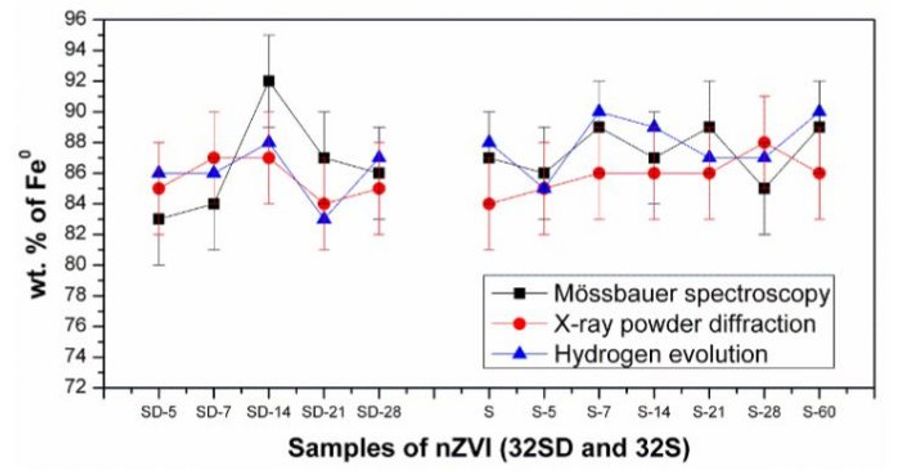
The 'Hydrogen method' has been compared to Mossbauer spectroscopy and X-ray powder diffraction - please see the following chart:
Reaction of nZVI and the acid proceeds according t...
Reaction of nZVI and the acid proceeds according to the formula:
2KHSO4 + Fe -> FeSO4 + K2SO4 + H2
55,85 g Fe-> 22,41 dm3 H2
From the previous formula it comes through, that 22,41 dm of hydrogen is generated by reaction of 55,85 g of iron and sufficient amount of acid (KHS04). The weight and the concentration of nZVI in slurry is consequently calculated from the volume of evolved hydrogen.
Customer reviews
No reviews were found for nZVI Tester - Device for Measurement of nZVI Content in a Slurry. Be the first to review!