

- Home
- Companies
- CloudLIMS
- Applications
- Laboratory Information Management ...
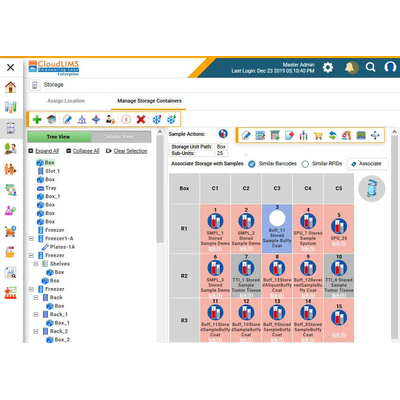
Laboratory Information Management System (LIMS) for Cloud Biobanking - Monitoring and Testing - Laboratory Equipment
Biobanks face numerous challenges, including managing biospecimens, associated metadata, and patient consent, locating stored samples, standardizing operational workflows, ensuring sample integrity, preventing unauthorized access to sensitive data, and following regulatory guidelines such as ICMR’s National Ethical Guidelines for Biomedical and Health Research Involving Human Participants 2017, Department of Biotechnology (DBT) guidelines, ISO 20387:2018, EU GDPR, 21 CFR Part 11, and ISBER Best Practices. CloudLIMS helps in securely managing sample and patient data, assigning role-based access to authorized users, following regulatory guidelines, removing data silos, and generating personalized reports. Being hosted in the cloud, Our biobanking LIMS helps in minimizing operational costs for biorepositories and in ensuring data integrity and security.
CloudLIMS is a purpose-built, scalable, SaaS, in the cloud biobanking LIMS and grows with your biorepository by supporting your data management, automation, and regulatory requirements. With a cloud solution, Our LIMS software for biorepositories can be accessed anytime, anywhere, is easy to deploy and enables biorepositories to quickly automate without the need to invest in IT infrastructure or personnel.
- Automated workflows to meet compliance and standards, such as ICMR’s National Ethical Guidelines for Biomedical and Health Research Involving Human Participants 2017, Department of Biotechnology (DBT) guidelines, ISO 20387:2018, EU GDPR, 21 CFR Part 11, and ISBER Best Practices.
- A flexible data model to configure and accommodate specific requirements and avoid costly customization.
- Customizable modules for managing samples and their genealogy tracking sample locations freezer inventory and managing shipment of samples when working with collaborators.
- Maintain a Chain of Custody to regulate the internal or external transfer of biospecimens from one custodian to another there by meeting compliance.
- Assign role-based data access to staff preventing unauthorized access and maintaining PHI (Protected Health Information).
- Manage studies and establish dynamic relationships between attributes across multiple modules and support simple to complex searches across multiple attributes.
- Support organizational standards with Single Sign-On streamlining user access to multiple applications and ensuring a complete and secure audit trail to meet regulatory requirements.
- Manage tests and results for clinical and research use and generate reports based on reporting requirements.
- Manage catalogs for sample purchase, storage, and testing services along with generating invoices.
