
- Home
- Companies
- LuminUltra Technologies Ltd.
- Applications
- Microbial testing for personal care ...

Microbial testing for personal care products sector - Health Care
The Problem: Microorganisms. personalcareMicrobiological contamination is a threat to product quality and process integrity in the personal care products and life sciences manufacturing industries – the purity of raw materials including water is essential to ensure manufacturing compliance. Microorganisms may be introduced at several stages of the process; subsequently, manufacturers must put in place stringent Quality Assurance / Quality Control (QA/QC) programs to manage microbiological contamination.In most cases, manufacturers rely on traditional culture testing (e.g. dip slides, plate counts) to monitor for microbiological threats in raw materials, intermediates, final products, and water systems. These tests require a 2-to-5 day incubation period. Batch release must await these results, and even then there is always a risk of undetected contamination if final products are released prior to firm confirmation of microbiological contamination control.
The cost of return for exchange of the compromised product can be many times the cost of identifying this contamination at the plant and represents the worst case, as not only are costs increased but also reputation is compromised. Therefore, manufacturers will benefit from having a way to more proactively identify samples collected from its manufacturing process for microbiological contamination.
QGO-M_sm2nd Generation ATP monitoring technology from LuminUltra offers a revolutionary yet affordable approach to microbiological quantification. Adenosine Triphosphate (ATP) is the common energy carrier for all forms of life, and as such its quantity relates directly to the amount of active, living biomass in a given sample. Through a line of test kits optimized for different applications and sample types, this technology provides quantitative, objective information on total microbiological population characteristics in 5 minutes or less.
2nd Generation ATP technology can be leveraged to augment your current microbiological monitoring and control practices. In other words, it is an additional test to be used in the QA/QC process to improve control practices. This is because ATP tests do not measure microorganisms in exactly the same way as traditional culture tests. Whereas culture tests measure only the microorganisms that are of the correct species and are in a culturable or viable state, ATP tests measure all organisms – that is, all species and all states. As such, ATP tests provide a more rapid and yet more conservative quantification of microbiological management initiatives.
Many FDA-regulated industries, such as the production of food and beverages, already make use of ATP measurement systems to shorten production cycles and reduce risk. LuminUltra`s products can bring the same time, cost and accuracy benefits to manufacturing industries such as:
- Pharmaceuticals
- Biotechnology
- Medical Devices
- Cosmetics
- Personal Care Products
ATP measurement is the #1 method for rapid identification and control of total microorganisms in the food & beverage manufacturing arena. Such techniques have saved manufacturers vast sums of money and resources by shortening production cycles, reducing recalls, and mitigating contamination risks. However, conventional `1st Generation` ATP measurement products are limited to surface testing, and cannot reliably detect microbial growth at low concentrations as required by personal care & life science manufacturers.
LuminUltra has bridged this gap with its 2nd generation technology, providing an ATP test kit that is fast, sensitive, accurate and inexpensive enough to finally provide operators with the same benefits for testing water and chemical products. LuminUltra’s line of test kits is flexible enough to be used in the following areas:
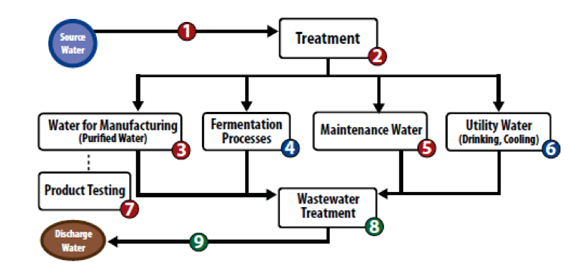
Source Water – Use the QGA™ test kit to measure microorganisms entering the plant to pace treatment requirements.
Water Treatment – Use QGA to monitor treatment efficiency and optimize maintenance practices.
Water for GMP/GLP Manufacturing – Use QGA to gain real-time feedback on microbiological contamination to reduce risk and shorten production cycles.
Fermentation Processes – Accurately measure biomass to optimize bioreactor operational parameters such as nutrient feed and batch cycle times using the QG21W™ test kit.
Maintenance Water – Use QGA to assess wash water cleanliness to avoid equipment contamination.
Utility Water – Use QGA to optimize microbial growth control programs for ancillary water systems (i.e. potable, cooling water).
Product Testing – Choose QGOM-XLPD™ (for filterable products) or QG21S™ (for non-filterable products) to monitor for microbiological contamination in final products and shorten quarantine cycles.
Wastewater Treatment – monitor biomass via QG21W in wastewater treatment processes to ensure regulatory compliance.
Discharge Water – monitor and manage effluent to meet regulatory compliance requirements (DEP/EPA).
LuminUltra has performed a validation of its QGO-M XLPD test kit for various personal care products.
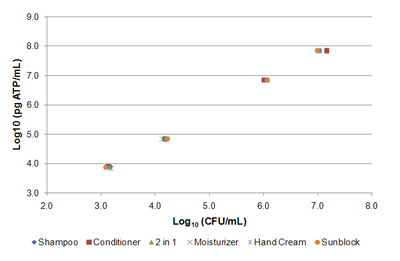
Comparison to Plate Counts
A comparison of ATP monitoring to standard heterotrophic plate counts for a mixed aerobic culture spiked in various products yielded a linear relationship between fg ATP/mL and CFU/mL (see chart).
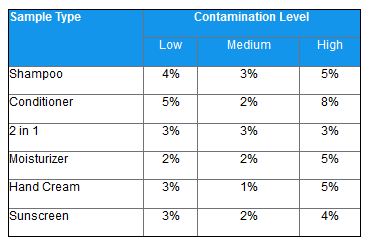
Excellent repeatability was demonstrated for triplicate measurements of ATP in different spiked samples of several personal care products (see table below).
Table 1: Repeatability Under Varying Contamination Levels
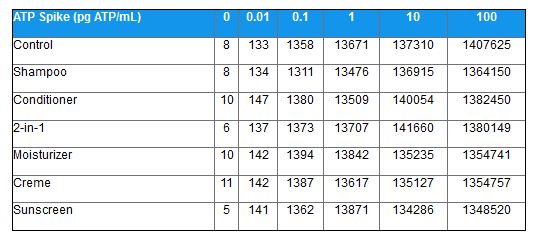
Finally, samples were spiked with known concentrations of ATP and the response (relative light units) was measured relative to a control (water). The results are shown in the table below and demonstrate no significant matrix effects for any sample type.
Table 2: Linearity Test Results in Several Personal Care Products
The costs associated with uncontrolled microbiological contamination in product manufacturing can be extremely high. Effective control starts with proactive, quantitative monitoring, which is best executed by rapid test methods. 2nd Generation ATP monitoring provides the most complete and rapid feedback for microbiological management on the market.
This technology can provide you with positive identification of microbiological contamination at the earliest possible juncture in any sample type. Consider the fact that a standard microorganism at slightly below room temperature will multiple (double) every four hours. This means that during the 2-day incubation period for a standard culture test, a sample that is already contaminated beyond control thresholds will double in size 12 times before a positive test result is known.
Furthermore, you can achieve prompt positive release of batches as soon as possible after filling is completed. This increases efficiency in operations and reduces inventory costs while assuring that microbiological quality specifications are met.
The potential payback associated with proactive microbiological monitoring is in the many tens or hundreds of thousands of dollars per year, realized through improved quality, increased production and reduced recall frequencies.
