- Home
- Companies
- CL Solutions, LLC
- Articles
- Fast BTEX and methylnaphthalene ...

Fast BTEX and methylnaphthalene bioremediation
Petrox Petrox and Methylnaphthalene Bioremediation Site Summary
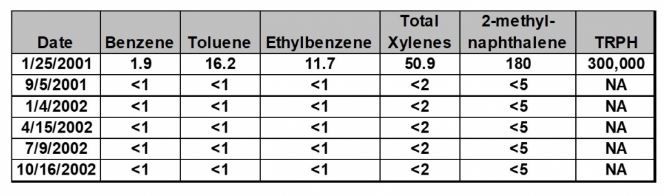
Petrox micorbes accelerated BTEX and methylnaphthalene bioremediation at a former fueling station in Florida. After a leaking tank underground storage tank (UST) was removed Petrox micorbes were applied to the groundwater. A temporary well showed high concentrations of BTEX, methylnaphthalene isomers, and total recoverable petroleum hydrocarbons (TRPH) in the former tank location. The temporary well was removed and replaced with a permanent monitoring point. Since the contaminants were not detected outside of the UST cavity, the treatment focused on the cavity and its proximity. Petrox was introduced into the ground water through 12 direct-push injection points in and around the UST cavity. Approximately 10 gallons of Petrox solution was injected through each of the injection points in August 2001. After one injection of Petrox, petroleum hydrocarbons were no longer detected in the UST cavity. Please refer to the table below for the ground water monitoring results.
Persistence in Soil and Low Ground Water Concentrations
Methylnaphthalene and naphthalene persist in soil after other volatile components of fuels are gone. The persistence is due to their relatively high affinity for adsorption to soil and relatively low water solubility. These factors account for the common rebound of ground water concentrations. When high concentrations of naphthalene and methylnaphthalene are present in soil near the ground water table, often the dissolved concentration in the ground water is very low. The accumulation of these compounds at the water table causes a problem for remediation. Frequently after ground water is remediated, seasonal fluctuation of the water table through the contaminated vadose zone recharges the concentrations of these compounds. The result is seasonal fluctuation in the ground water concentrations.
The micorbes in Petrox accelerate the remediation because they product an extracellular biosurfactant that desorbs the methylnaphthalene to make it available for extraction or for in situ bioremediation. At this site, the contaminated soil was removed to the water table, so concentrations did not rebound after the initial ground water remediation. At other sites where Petrox bioremediation was used to address methylnaphthalene or naphthalene, the bioremediation was combined with extraction. The combined technologies removed the contamination flushed from the soil in addition to the bioremediation.