

Hard to Find Filter Configurations
Announcing New Hard to Find Filters
Filters have been the go-to method for sampling air in determining particulate contamination.
Before I get into the “hard to find” filters, let us talk about filtration.
The ideal filter should:
- be able to capture “everything” down to the nanometer;
- never plug;
- be free of the substance that you are measuring, down to nanograms;
- contribute zero pressure drop when “clean”, at the operating flow rate;
- have trivial cost;
Well no such filter exists, but with careful planning and sourcing, you can come close to fulfilling your requirements.
Point 1. Capture Everything?
Most filters on the market have their pore size rated in micrometers (µm) a.k.a. microns. That implies their ability to capture particulates larger than their pore size. This viewpoint, also erroneously implies that anything smaller, would go through the filter.
What is not commonly known, is that their ratings were arrived at, decades ago, when filters were primarily used to remove particulates from liquids, not air.
Filters were rated with a 98% nominal retention of particles larger than their rated pore size. For example, think of the old standby What man filters used in your freshman chemistry class. Those filters did not even have a micron rating attached to them.
As membrane filters came into use in the early 1950’s, ratings were attached to them and expressed in micrometers. Again, most applications were to clarify liquids. Consequently, the ratings were determined by experiment using liquids as the medium, only now, a stricter rating of absolute µm was given, which implied 100% retention of particles greater than the rated pore size. To put this concept across, membrane filter manufactures conjured a visualization aid — the sieve.
Although the sieve helped make the point, it also gave the wrong impression that particles smaller than the pore size, would pretty much, go 100% through the filter. A scientific paper, shows the extent of this misrepresentation. (Retention of Marine Particles by Screen and Filters Sheldon & Sutcliffe 1992 Limnology and Oceanography pp 411-414. )
As air pollution, both indoor and outdoor, became a health concern, membrane filters were used to capture airborne particulates for quantitative detection of various dusts, i.e., total dust, metal dusts, silica, coal.
Unfortunately, the retention ratings that were assigned to membrane filters (using liquid filtration to rate them) are meaningless and misleading when it comes to the choosing appropriate filters for sampling of air.
Stark Differences Between Liquids and Air !
In liquids, the viscous drag on particles that are trapped on the filter is enormous. Any particle smaller than the pore size is subjected to forces that try to drag the particle through the filter’s maze of pathways.
Compare that to air which is at least 3 orders of magnitude less viscous. As a result, the retention capability of a filter used to filter a gas, is greatly magnified and makes the filter far more likely to retain smaller particles, through the mechanism of entanglement, and impaction.
The typical membrane is about 85-125 microns thick irrespective of its pore size.
So, a one micrometer particle has a long way to go to traverse the filter.
Most membrane filters are made of plastic, they tend to build up electrostatic forces, which can attract and hold particles within the pore structure. This static contributes to another force that tends to increase the efficiency of retention of particles below the rated pore size. In liquids this static force is quickly dissipated through contact with the liquid.
In an experiment by Wiffen and McGaw in 1959
(The Efficiency of Membrane Filters J. Air Water pollution 1963 Vol 7 pp 501-09), a 0.8 µm membrane filter (rated as 0.8 µm via liquid filtration) retained 50% of 0.008 µm (8 nanometers) particles at a pressure drop of about 12 psi. (A filter used in the vacuum mode, at sea level, exerts a maximum of 14.3 lbs. of pressure across the filter).
If one examines the % retention curve, shown in the Wiffen McGaw paper, it is estimated that the 98% retention occurs somewhere around 0.08 µm. (This point is important as I will explain later.)
The above experiment shows that membrane filters, when used with vacuum to sample air, are extremely efficient, well below their liquid rated micrometer rating.
Point 2. Never Plug?
Well you can never have a filter that never plugs, all filters, in the real world have a certain “dirt holding” capacity, before they become useless due to an impractically low flow rate. With a bit of experimentation, you can empirically arrive at a compromise, that balances the practicality of sampling in a short enough time and the need to capture enough contaminant to be able to analyze accurately with a low detection limit, for the contaminant in question. That is where understanding the impact of what I described in Point 1. Capture Everything? You are no longer limited to choosing a filter that has a micrometer rating below the smallest particle you wish to capture.
Let me illustrate with the following example:
Extrapolating from the data generated in the Wiffen & McGaw paper, if you choose a 5.0 µm liquid rated filter: you will capture 98% of 0.05 µm particles. That is two orders of magnitude smaller diameter than the liquid rated poresize.
Well you may say, that is not good enough, I want to get closer to 100% capture.
OK, but let me explain why in the real world that would not be necessary.
If we make some more assumptions:
1. Assume most particles are close to a spherical shape.
(In the Wiffen McGaw paper, the particles used were close to cubic shape.
Spherical particles are the ones that would have the properties for the best penetration into and through a filter. Fibers and other particles shaped between a sphere and a needle would have decreasing penetration.)
- A sphere has a volume of V= 4/3 πr3, so a 0.05 µm particle would have a volume of 0.00016667 µm3
- Assume a log normal distribution of particles in the atmosphere sampled
- Assume the sample collected, has a distribution as follows in the chart below
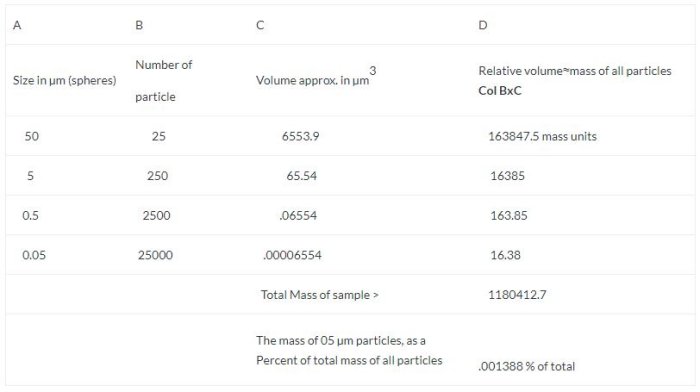
Putting it in another way, although 2% of the particles captured went through the filter, they only amounted to .001388% of the total mass. An insignificant quantity.
As you can see, using a “coarser” filter as a capture medium for any analytical method ( gravimetric, ICP, AAS HPLC, etc.) that is based on the mass of the contaminant to determine the quantity, will not be affected by the loss of such a negligible amount.
Check our blog next week to read more about points 3, 4, and 5, of the “hard-to-find” filters discussion.
