Piper Diagrams in Chemistry Water Analysis
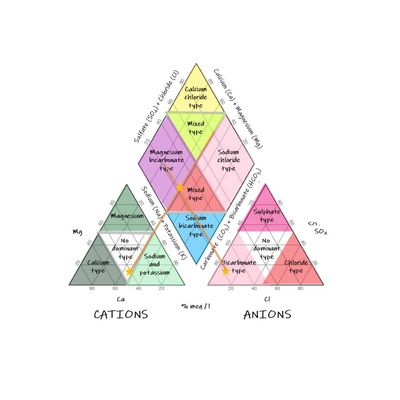
Arthur M. Piper introduced the Piper diagram in 1994. It is used for studying water chemistry and quality. The Piper diagram helps in the segregation of analytical data necessary for determining the sources of the constituents dissolved in water. The applicability of the procedure is possible due to natural water having anions and cations in chemical equilibrium. The most abundant cations in water are Calcium (Ca), Magnesium (Mg) and Sodium (Na) while the anions are bicarbonate (HCO3), sulphate (SO4) and chloride (Cl). According to Piper (1994), the diagram is made by drawing two triangles which correlate to cations and anions. It also includes a diamond which is used in summarizing the two triangles, as shown below.
Piper diagrams are used by hydrogeologists and groundwater analysts to assess the composition of cations and anions and therefore determine the sources of water. The tool is useful in developing an understanding of the chemistry of water through analysis. Hydrochemistry is a field which positively benefits from the use of Piper diagram as it provides insights on hydrochemical processes. These processes are influenced by cation and anion exchanges, dissolution of minerals, and silicate weathering.
Additionally, through water quality analysis, water use is determined depending on its quality. For instance, water can be used for agricultural activities, drinking and industrial purposes. It is worth noting that cation and anion concentration in water defines its properties and thus ways in which it can be used. For instance, residual sodium carbonate defines the suitability of water for irrigation purposes. Generally, Piper diagram is used for water analysis to develop an understanding of water sources and properties and therefore define water use purposes.
Piper diagrams can be produced using environmental data management software. ESdat, for example, uses a modification of an Excel based piper diagram developed by the United States Geological Survey (USGS). For more information https://esdat.net/ESdatHelp/index.html?piper.htm
References
Kumar, P.S.J. (2013). Interpretation of groundwater chemistry using piper and chadha´s diagrams: A comparative study from Perambalur Taluk. Elixir Geoscience, 54, 12208-12211.
Montoya, S. (2018). What is a Piper diagram for water chemistry analysis and how to create one? Retrieved from https://www.hatarilabs.com/ih-en/what-is-a-piper-diagram-and-how-to-create-one