Focus Laboratories Inc. products
Pharmaceutical Microbiology - USP 61 Nonsterile Product Microbial Enumeration
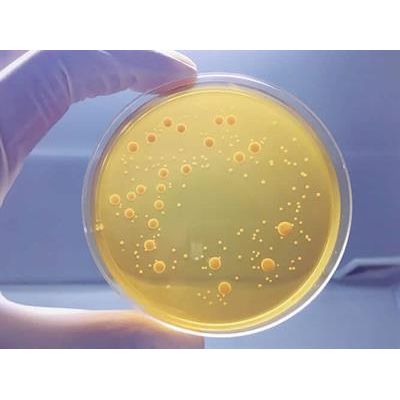
Focus - Aerobic and Yeast and Mold Plate Count
10 gm of sample is diluted with a pH balanced, isotonic diluent, usually supplemented with chemical neutralizers for any antimicrobial agents which might be present in the drug product, and further homogenized by shaking, stomaching, vortexing or other means.
Focus - Membrane Filtration
Sample prepared in diluent as in Pour Plate or Spread plate but the resulting dilution is passed through a 0.22 micron filter. The filter is removed and placed on the surface of nutrient agar plates, allowing microbes to growth to visible colony forming units. Membrane filtration is recommended with chemical neutralization of antimicrobial agents is not possible, such as with antibiotics. The filtration physically separates the antimicrobial from the contaminant, leaving the microbe on the surface of the filter for further culturing. It is also useful when the limit of detection must be
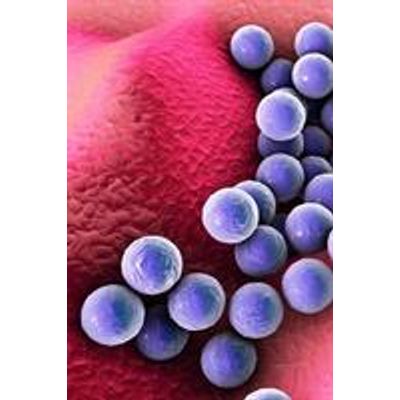
Pharmaceutical Microbiology - USP 62 Nonsterile Product Test for Specified Microorganisms
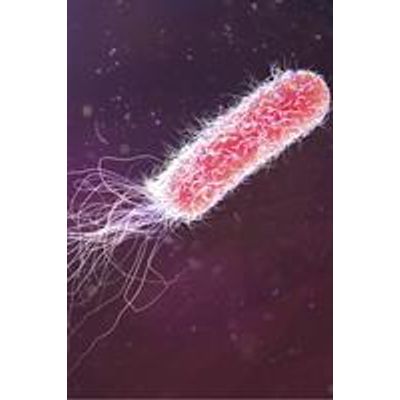
Focus - Pseudomonas Aeruginosa
A Gram negative aerobic rod, P. aeruginosa is an opportunistic pathogen, typically infecting the pulmonary system, urinary tract, burns and wounds. It is a ubiquitous microbe, found in soil, water, and on skin.
Focus - Escherichia Coli
E. coli is well known as a pathogen, it is often in the news causing outbreaks of food poisoning. It is a Gram negative, facultative anaerobic coliform, commonly found in the intestine of mammals and birds. Most strains are harmless, but E. coli is also one of the most diverse species of bacteria, and those strains that produce shiga toxin are particularly lethal. The important point regarding E. coli is that it is a coliform, or fecal contaminant, and clearly indicates that the drug product that was not made under GMP conditions.
Environmental Monitoring - USP 1116 and ISO 14698
Focus - Alert and Action Levels
Many Life Science Industries base their EM programs on USPMicrobiological Control and Monitoring of Aseptic Processing Environments or ISO 14698 Cleanrooms and associated controlled environments – Biocontamination control.
Medical Devices
Focus - Environmental Monitoring Devices
FOCUS Laboratories can help develop an environmental monitoring program that is useful form medical device manufacturers. Utilizing principles outlined in AAMI TIR52 Environmental Monitoring for Terminally Sterilized Healthcare Products, FOCUS Laboratories can help determine sample sites, sample frequency, methodology as well as interpret results. For more information on our Environmental Monitoring capability
Focus - Bioburden Viable Microbes
The amount and type of viable microbes on a medical device must be known to set adequate sterilization measures. Since microbial contamination can be sporadic, bioburden is determined on replicate lots of product, with replicate units from each lot. Typically a medical device is submerged in a rinse fluid and shaken, stomached, sonicated or treated in some other fashion that will rinse the microbial off the device. The rinse is then evaluated for microbial bioburden, according to methods
Focus - Endotoxin
If the device is rinsed with water at any point in the manufacturing process, there is a high likelihood of endotoxin contamination. This endotoxin contamination will not be remedied with post process sterilization. Water used in medical device manufacture should be tested for endotoxin, as well as the medical device itself.