- Home
- Companies
- Microbiologics
- Products
Microbiologics products
Cannabis Microbial Testings
Epower - Model CRM - Quantitative Quality Control Microorganisms Product
The ultimate in authenticity. Epower CRM quantitative quality control microorganisms are just one passage from the reference strain. If your lab is ISO 17025 accredited, Epower CRM can help you meet the requirements of Section 5.6.3.2 regarding reference materials. This product offers plenty of flexibility, which is a huge benefit in the lab. Each microorganism pellet has a pre-determined range of CFUs, so you can easily manipulate the product to deliver your desired concentration. You can also create a mixed microorganism population by combining multiple Epower CRM strains. As a Certified Reference Material, Epower CRM comes with a comprehensive Certificate of Analysis detailing the strains identity, characteristics and standard deviation.
Microbiologics - Model ANA (3 Strains) QC Set - QC Sets and Panels
Clinical
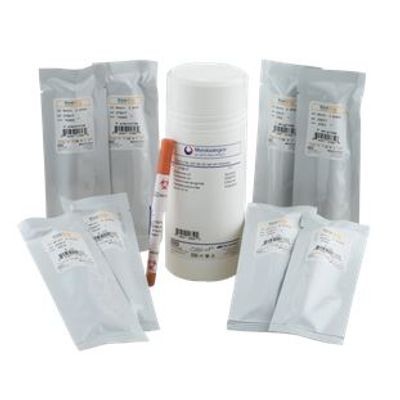
Lyfo Disk - Qualitative Microorganism Pellets
It doesn’t get much easier than this. Simply rehydrate LYFO DISK qualitative microorganism pellets with a diluent, such as phosphate buffer or saline, and you’re ready to inoculate your culture media. LYFO DISK, packaged in a glass vial containing 6 pellets of a single strain, offers a ton of flexibility. It`s an ideal solution for many quality control applications including presence/absence testing, microbial identification methods, antimicrobial susceptibility testing, media QC, water testing and more!
Education
Kwik-Stik - Qualitative Lyophilized Microorganism Pellet
Laboratories throughout the world have been praising the KWIK-STIK for decades. That’s because its simple, all-inclusive design makes life easier for lab techs while reducing chances for errors. Each KWIK-STIK contains a qualitative lyophilized microorganism pellet, ampoule of hydrating fluid and inoculating swab. Everything you need to grow reference cultures for QC testing is included in this one handy device. Available in packs of 2 or 6, KWIK-STIKs are universally used for all types of microbiological control testing.
Food Safety Microbial Testing
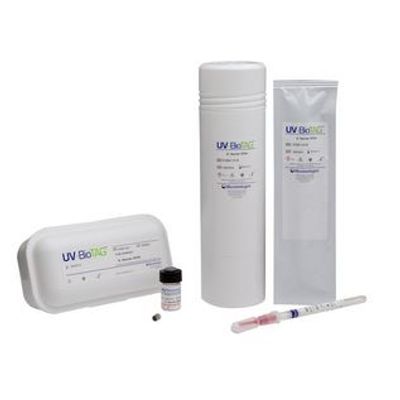
Microbiologics - Model UV-BioTAG - Green Fluorescent Protein (GFP)
It’s not uncommon for food samples to inadvertently become contaminated with control strains used in the laboratory. This leads to false-positive results which can have severe financial and regulatory implications. UV-BioTAG, our line of reference strains containing green fluorescent protein (GFP) markers, make it easy to distinguish standard microorganism strains from laboratory control strains. Optimized for highly visible fluorescence, UV-BioTAG strains provide better stability because the GFP marker is integrated into the chromosome rather than the plasmid. With UV-BioTAG, you can quickly and reliably establish whether a positive test result can be traced back to a control strain cross-contamination issue, or if it is from another source.
Pharmaceutical
Microbiologics - Model EZ-Accu Shot Select - Convenient Kit
EZ-Accu Shot Select contains the 5 compendial organisms for Growth Promotion Testing,plusE. colifor a total of 6 strains in one convenient kit. Perfect for qualifying EZ-Accu Shot for use in your laboratory, the EZ-Accu Shot Select kit contains one instant dissolve pellet of each of the 6 strains and 6 vials of hydration fluid. Each pellet delivers 10-100 CFU per inoculum with no serial dilutions and provided 8 hours of stability after rehydration for added flexibility to perform tests. EZ-Accu Shot Select strains are just 3 passages from the reference culture to meet Pharmacopeia standards.
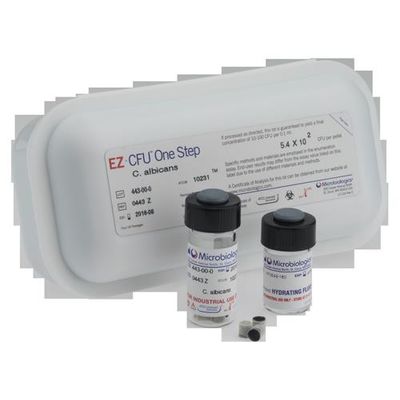
Microbiologics - Model EZ-CFU One Step - Ready-to-Use Quantitative QC Microorganisms for Growth Promotion Testing
Is your lab still preparing microorganism suspensions the old fashioned way for Growth Promotion Testing of culture media? Allow us to introduce you to your new favorite lab supply, EZ-CFU One Step. We designed this product specifically for Growth Promotion Testing, Suitability of Tests for Specified Microorganisms, Suitability of Counting Methods and more. These ready-to-use quantitative microorganisms are just 3 passages from the reference culture and deliver 10-100 CFU per inoculum, as required by the Pharmacopeias, with minimal prep time.