

- Home
- Companies
- Bioquell PLC, an Ecolab Solution
- Products
- Bioquell - Bioquell Integrated Building ...
Bioquell - Bioquell Integrated Building Decontamination System for Facilities
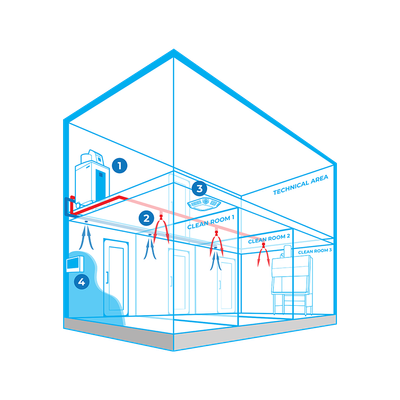
The Bioquell Integrated Building Decontamination System is engineered for efficient and routine decontamination of specific areas within facilities, such as containment labs or sterile manufacturing cores. At its core, the system employs centrally located hydrogen peroxide vapor generators that distribute the vapor through a building's existing ventilation system or a dedicated network of pipes. This configuration minimizes manual setup, enhances repeatability, and reduces human error. Ideal for biosafety and biomedical environments, this automated system can integrate with Building Management Systems. It allows for multiple decontamination cycles, ensuring a high efficacy by achieving a 6-log sporicidal kill. Bioquell prioritizes safety and efficiency, ensuring that the equipment's location in unclassified areas simplifies maintenance and prevents personnel exposure to contamination. This system is optimal for stringent decontamination needs in pharmaceutical and biotechnology operations, ensuring continued compliance and operational safety.
The Bioquell Integrated Building Decontamination System is a fully integrated solution designed for frequent or routine decontamination of the same area within a facility. This could be anything from a containment lab or multiple material airlocks, to a sterile manufacturing core or biomedical holding area.
Typically, Bioquell hydrogen peroxide vapor generator(s) are located centrally in a technical or unclassified space, and the vapor is distributed to the areas where it is required either through the building’s ventilation system or a dedicated network of pipes leading to nozzles on the ceiling of the target rooms. With an integrated system, no movement or storage of mobile equipment is required reducing manual setup times, increasing repeatability and removing human error.
- GMP Biopharmaceutical manufacturing areas
- GMP Filling suites
- Biomedical facilities
- Biosafety/containment laboratories
Efficient
No mobile equipment to store or set up in classified area meaning no trailing cables or sealing of doors is required, removing human error
Effective Distribution
Vapor can be distributed either using the building’s ventilation system or through a dedicated network of pipes
Automated
Can integrate with BMS and utilize actuated valves to control the delivery of vapor into each individual room, allowing a number of different decontamination cycles to be run at the touch of a button
Rapid
Quicker setup times, faster decontamination cycles and the ability to run the system overnight means less downtime
Efficacious
Validated and programmed cycles ensure repeatable 6-log sporicidal kill with Bioquell Hydrogen Peroxide Sterilant (EPA Registration Number: 72372-1-86703)
Safe
With the equipment located in an unclassified area, service and maintenance is easier and no personnel entry is required into a potentially contaminated target area
