Micro-Media - Model L Series -Depth Filter Media
Micro-Media L Series Depth Filter Media, is specifically designed for use in applications where proteins are present, such as blood fractionation and recombinant DNA derived products. These particular types of applications demand specific controls on pyrogens and extractables. Physical attributes, unique to such applications can lower the surface tension of the media and cause them to release extractables, which can cause problems with the LAL testing used both in process and as a final release. These extractables can cause “false positives” by reacting with a Factor G in the LAL reagent and cellulose components.
A particular source of extractables is 1,3 ß D-glucan contamination in the cellulose. Cellulose is a major component of depth filter products. The level of reactivity depends upon the amount of 1,3 ß D-glucans released during manufacturing of the filter media and the amount of Factor G present in the LAL reagent, which varies by manufacturer.
Utilizing a unique process, ErtelAlsop minimizes 1,3 ß D-glucan levels in cellulose pulp prior to manufacturing the filter media. This minimizes the effects of 1,3 ß D-glucans and allows for validation of the LAL test on the product.
Composition
Micro-Media L Series Filter Media is available in sheets or Pak Lenticular Cartridges. Micro-Media Filter Pads provide enhanced levels of particulate removal due to strong electrokinetic forces, which adsorb particles smaller than the media nominal pore size rating. Pharmaceutical Grad L Series Media is manufactured in accordance with ErtelAlsop’s Drug Master File at CDER.
- Blood Fractions
- Recombinant DNA Derived Products
- Sera
- Cell Culture Media
- Active Pharmaceutical Ingredients
- Large Volume Parenterals
- Small Volume Parenterals
Product testing is always available either at your facility, through our network of distributors, or at our in-house laboratory.
Pilot Testing - Rentals
For in-plant testing and scale-up procedures, ErtelAlsop offers a variety of lab filters for rent. A portion of rental fees can be applied to the purchase of your full production filter.
The most accurate way to optimize your process is through laboratory scale testing. Samples of Micro-Media L or Micro-Clear™ L Series Media are available at no charge. Authorized ErtelAlsop representatives are equipped to run trials on-site, or product samples may be sent directly to ErtelAlsop for testing with prior approval.
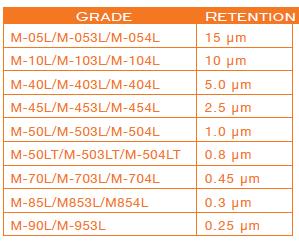
Table to the left is for reference only.