

- Home
- Companies
- Dyadic International, Inc.
- Products
- Model C1-Cell - C1-cell Protein ...
Model C1-Cell -C1-cell Protein Production Platform for Drug Development
Our lead technology, C1-cell protein production platform, is based on an industrially proven microorganism (named C1), which is currently used to speed development, lower production costs, and improve performance of biologic vaccines and drugs at flexible commercial scales for the human and animal health markets. Dyadic’s naturally derived C1 cells went through random mutagenesis. This development brought about highly beneficial changes and transformed the shape of the C1 cells, from long strands to short, grain-sized sections.

C1’s unique morphology, its novel form and structure resulted in higher productivity and better growth conditions leading to the manufacture of low-cost biological products. Over the past two decades, scientists have made various additional genetic modifications to C1 cells leading to further increases in productivity, purity, stability, and quality. C1 cells don’t sporulate during manufacturing and can be grown on low-cost synthetic media at scales ranging from microtiter plates, single use bioreactors to large industrial scale stainless steel fermentation tanks.

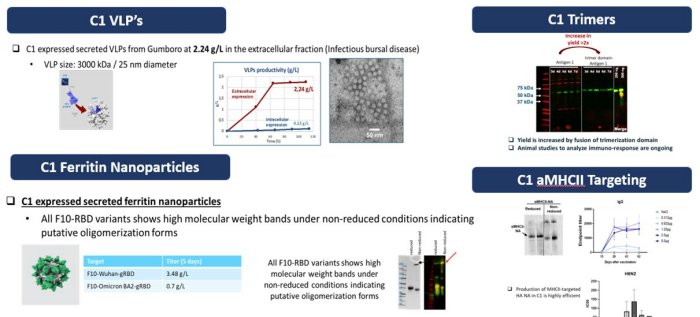
C1 can express diverse classes and types of biological molecules, including recombinant enzymes, antigens, ferritin nanoparticles, virus like particles, and therapeutic proteins, such as Fc-fusion proteins, and monoclonal and bi-specific antibodies.
Meeting the growing demand for proteins worldwide for human and animal health with highly productive scalable microbial biomanufacturing platforms and products.
To improve how we feed, fuel, and heal the world by utilizing modern biotechnology to revolutionize medicine, agriculture, nutrition, and food.
Current ChallengesCurrent methods to produce high quality and high throughput proteins at low cost are not adequate to meet the growing global demand for vaccines, monoclonal and other antibodies, therapeutic proteins, as well as food, nutrition, and diagnostic enzymes and proteins.
The pandemic has highlighted key challenges that have existed within biomanufacturing that will only increase as demand or circumstances, such as a global pandemic or endemic threat, climate change, and population increase continue to drive rising demand not only for lifesaving biologic products but also food and nutrition products.
Shortened timeframes and shortage of raw materials, equipment, consumables, lipid nanoparticles due to high demand
Examples: Cell culture media, Bioreactor bags, vials, tangential flow filters
Inherent challenges include time to produce new cell lines, drug substance, process optimization, aseptic techniques, cold manufacturing
Infrastructure and production requirements, cold storage and distribution challenges, geographic challenges
- Stable Cell lines in ~7 weeks
- Research Cell Bank becomes Master Cell Bank
- No viral or endotoxin inactivation
- 3-7 day fermentation (continuous in development)
- Up to 300 times more productive than certain currently used vaccine cell lines
- Up to 10 times more productive than certain currently used mammalian cell lines
- Industrially proven platform that has run at up to 500,000L
- Record levels of 10g/L of an antigen, >22 g/L of a monoclonal antibody
- Industrial scale flexibility and scale
- C1 can lower CAPEX:
- Produce at smaller scale while dramatically increasing protein yields
- Unparallelled productivity provides margin relief
- C1 can lower OPEX
- Smaller facility footprint and related costs
- Industrial media costs for pharma grade materials
