Pharmigene, Inc.
- Home
- Companies
- Pharmigene, Inc.
- Products
- Pharmigene - Model HLA-B*5801 - RT-PCR ...

Pharmigene - Model HLA-B*5801 -RT-PCR & SYBR Green Based Detection Kit
FromPharmigene, Inc.
RT-PCR & SYBR Green based detection method: each sample requires only 2 PCR rxns for detecting of HLA-B*5801 and Internal Control. Analysis is based on Ct values. A complete kit version has IVD approval from Department of Health, Taiwan.
Most popular related searches
- Required only hours from blood sample to results.
- Designed for easy operation & analysis.
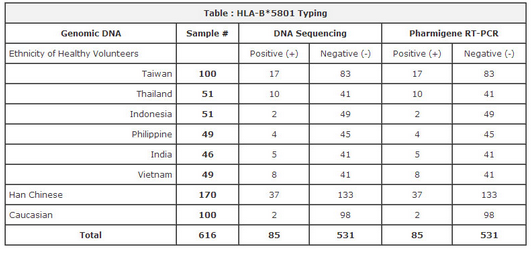
- Validated accuracy with 600+ patients samples* & DNA sequencing.
- Protected by US, Europe & world wide patents.
- Mix & Match design.
Regulatory Status
HLA-B*5801 Detection
- Taiwan IVD
- China IVD
- US ASR
Many studies have demonstrated the strong association of Human Leukocyte Antigen (HLA-B*5801) and allopurinol induced Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN). Individuals that use DNA tests to determine their HLA genotypes prior to taking these drugs can reduce their risks of developing SJS/TEN significantly.