

ResistancePlus - Model MG FleXible -Detect Mycoplasma Genitalium and Macrolide Resistance Markers
Detect Mycoplasma genitalium and macrolide resistance markers; Sample-to-answer solution validated for all Cepheid GeneXpert® Systems.
Mycoplasma genitalium (Mgen) infections contribute to significant portion of non-chlamydial non-gonococcal urethritis in men and in women and treatment is challenging due to high levels of macrolide resistance.
ResistancePlus® MG FleXible offers simultaneous detection and identification of M. genitalium and azithromycin (macrolide) resistance to support appropriate treatment decisions at the earliest opportunity for improved patient management.
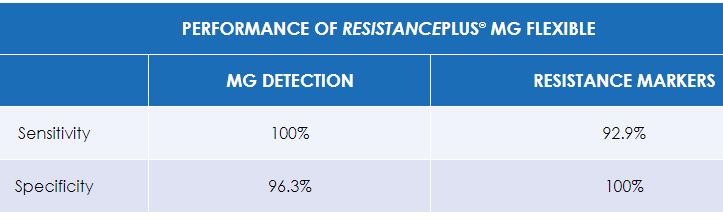
Validated on hundreds of clinical samples, ResistancePlus® MG FleXible demonstrates excellent sensitivity and specificity.
ResistancePlus® MG FleXible brings accuracy and simplicity to your lab in combination with GeneXpert® Systems for a complete solution. Easily implemented into your existing workflow or setting.
- Minimal preparation time ~ 10 mins
- Actionable results in around 2 hours
- Validated on a range of specimen types and collection devices
The ResistancePlus® MG FleXible assay is a qualitative multiplexed in vitro diagnostic real-time PCR test for the identification of M. genitalium and detection of mutations in the 23S rRNA gene, that are associated with resistance to azithromycin (macrolide antibiotic).
Single well: M. genitalium (MgPa), A2058G, A2059G, A2058T, A2058C, & Internal Control
Sample types- Urine: Male and female
- Swabs: vaginal, cervical, rectal, and urethral
- GeneXpert® Instrument Systems (Cepheid)
The components of the ResistancePlus® MG FleXible kit (P/N:2000410-R) are shipped on dry ice or ice gel packs and stored at -20°C. ResistancePlus® MG FleXible Cartridges (P/N:2000410-CART) are shipped and stored 2°C – 28°C.
Each shipment will include a ResistancePlus® MG FleXible CD (P/N:CD-MG-FLEX), containing Instructions for Use (IFU) and the Assay Definitiion File (ADF) needed to run the ResistancePlus® MG FleXible kit on the GeneXpert® Instrument.
Storage & stabilityExpiry dates are stated on the labels. It is recommended that freeze/thaw cycles be limited to less than 8. Store protected from light at – 20°C.
Intended useFor in vitro diagnostic use. Not for sale in the USA.
Regulatory statusCE-IVD (may not be available in all countries), TGA cleared
