Sunkaier - Methylene Diphenyl Diisocyanate (MDI) Technology
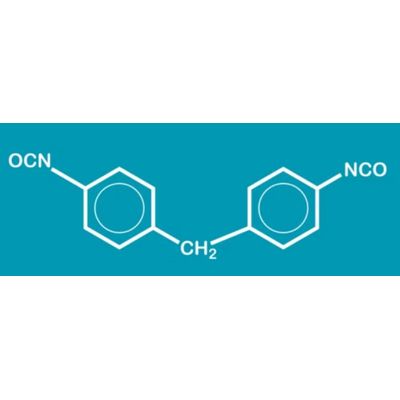
Methylene diphenyl diisocyanate (MDI) is an aromatic diisocyanate. Three isomers are common, varying by the positions of the isocyanate groups around the rings: 2,2′-MDI, 2,4′-MDI, and 4,4′-MDI.T he 4,4′ isomer is most widely used, and is also known as 4,4′-diphenylmethane diisocyanate.
1. Cold Phosgenation (Carbamoyl Chloride Formation)
MDA is dissolved in an inert solvent (e.g., chlorobenzene) and reacted with phosgene at low temperature (0–40°C)
2. Hot Phosgenation (Conversion to Isocyanate)
The intermediate is heated to 100–200°C to form MDI
3. HCl Removal & Purification
The crude MDI is distilled to remove solvents, HCl, and unreacted phosgene. The final product is a mixture of monomeric MDI (pure 4,4`-MDI, 2,4`-MDI) and polymeric MDI (PMDI).
- Construction: Insulation panels, spray foam, sealants
- Furniture & Bedding: Mattresses, sofa foam, cushions
- Automotive: Seats, dashboards, bumpers
- Footwear: Shoe soles, sports shoes
- Wood Composites: MDF. particleboard adhesives
- Industrial:Coatings, adhesives, elastomers
- Medical: Prosthetics, medical foams
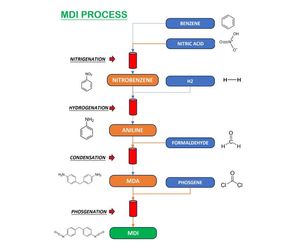
MDI is produced from Benzene by nitration to Nitrobenzene (NB) followed by hydrogenation of NB to produce Aniline(AN). The Aniline is condensed with formaldehyde to produce a diamine, MDA (Methylene diphenyl diamine). MDA is finally phosgenated to produce the end product, MDI. The crude MDI consists of several different isomers and oligomers which are separated into two ring MDI (pure MDI and OMDI) and oligomeric MDI’s, PMI.
Aniline
Aniline (CAS No. 62-53-3) is produced in a continuous high efficiency liquid phase hydrogenation process. The process utilizes a proprietary catalyst for maximum economy and selectivity.
MDA
Methylene dianiline (4,4-MDA,CAS No.101-77-9) and oligomeric MDA (PMA, CAS No. 25214-70-4) is produced by condensation of Aniline with formaldehyde in acidic environment. The overall process is continuous but it uses a combination of tank and batch reactors.
MDI
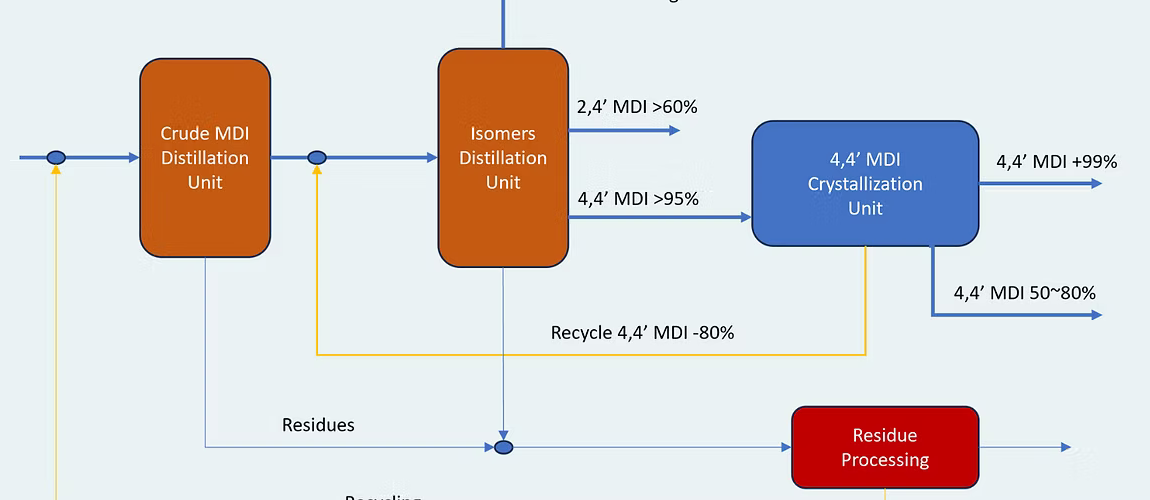
MDI Mixed isomers (MDI, CAS No. 26447-40-5) and oligomeric MDI (PMI, CAS No. 32055-14-4) is produced by reacting PMA with phosgene in a solvent. The solvent and the excess phosgene is recovered and recycled. The by-product HCI is purified and either oxidized to produce chlorine which is used in the phosgene synthesis or sent outside the plant area for use in e.g. a VCM plant. Part of the HCI gas is absorbed in water to give hydrochloric acid which is used in the MDA synthesis. The crude PMI mixture is further purified and separated into 2-ring MDI (CAS No. 26477-40-5) from which pure 4, 4’-MDI (CAS No.101-68-8) and a 2,4’-MDI rich fraction (OMDI, CAS No. 5873-54-1) is produced. The remaining oligomeric fraction is PMI and it is, together with the 2-ring material, either sold as it is or blended and / or modified into various MDI based products.
Methylenediphenyl Diisocyanate (MDI) production process including the synthesis of aniline, formaldehyde condensation, and phosgenation.
1. Benzene to Aniline
Aniline is first produced from benzene through a two-step process:
a) Nitration of Benzene
Benzene is nitrated using a mixture of concentrated nitric acid (HNO3) and sulfuric acid (H2SO4)to form nitrobenzene
b) Hydrogenation of Nitrobenzene
Nitrobenzene is then hydrogenated in the presence of a metal catalyst (e.g., Cu, Ni, or Pd) to produce aniline
2. Aniline-Formaldehyde Condensation (MDA Synthesis)
Aniline reacts with formaldehyde (HCHO) under acidic conditions to form Methylenedianiline (MDA), a mixture of isomers (mainly 4,4`-MDA, 2,4`-MDA, and 2,2`-MDA)
Catalyst: Typically HCl or H2SO4.
• Temperature: ~50–100°C.
3. Phosgenation of MDA to MDI
MDA is reacted with phosgene (COCl2) to produce MDI (Methylene Diphenyl Diisocyanate)