

- Home
- Companies
- MtoZ Biolabs
- Services
- MtoZ Biolabs - Host Cell Protein (HCP) ...

MtoZ Biolabs - Model hcp-antibody-coverage-analysis -Host Cell Protein (HCP) Antibody Coverage Analysis Service
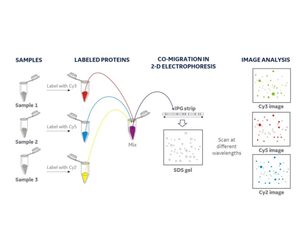
HCP antibody coverage refers to the proportion of specific host cell proteins (HCPs) detected by antibodies during pharmaceutical production. HCPs are protein impurities that inevitably remain in the final product during the production of recombinant proteins or monoclonal antibodies using host cells (such as E. coli, mammalian cells, etc.). These HCPs may affect the safety and efficacy of the drug, and therefore must be removed or reduced to acceptable levels through specific purification steps.
HCP antibody coverage analysis is based on the principle of antigen-antibody specific binding, by combining the HCPs in the sample with specific HCP antibodies, and then measuring the amount of the complex using other methods such as ELISA, 2D-DIGE, etc. By comparing with known concentrations of standards, the content of HCPs in the sample is determined. This process helps us understand how many different types of HCPs are recognized and detected by the antibodies, thereby evaluating the coverage of these antibodies.
Razzazi-Fazeli, E. et al. Aflatoxins - Biochemistry and Molecular Biology. 2011.
With the increasing scale and complexity of biopharmaceutical production, HCP antibody coverage analysis has become a key step in the quality control of biopharmaceutical products. Moreover, by assessing the purity and quality consistency of the product, this method has broad application value in the biopharmaceutical industry. The company has established four HCP antibody coverage analysis technology platforms combining regulations and practical applications, including 2D-WB, 2D-DIGE, AAE-2D, and AAE-nanoLC-MS/MS, to ensure the accuracy of HCP antibody validity analysis and meet various research needs. Free project evaluation, welcome to learn more details!
In the technical report, MtoZ Biolabs will provide you with detailed technical information, including:
1. Experimental Procedures
2. Relevant experimental Parameters
3. Detailed Information on HCP Antibody Coverage Analysis
4. Relevant Images
5. Raw Data
MtoZ Biolabs is an integrate contract research organization (CRO) providing advanced proteomics, metabolomics, bioinformatics, and biopharmaceutical analysis services to researchers in biochemistry, biotechnology, and biopharmaceutical fields. The name of MtoZ represents “mass to charge ratio” in mass spectrometry analysis, as most of our services are provided based on our well-established mass spectrometry platforms. Our services allow for the rapid and efficient development of research projects, including protein analysis, proteomics, and metabolomics programs.
MtoZ Biolabs is specialized in quantitative multiplexed proteomics and metabolomics applications through the establishment of state-of-the-art mass spectrometry platforms, coupled with high-performance liquid chromatography technology. We are committed to developing efficient, and effective tools for addressing core bioinformatics problems. With a continuing focus on quality, MtoZ Biolabs is well equipped to help you with your needs in proteomics, metabolomics, bioinformatics, and biopharmaceutical research. Our ultimate aim is to provide more rapid, high-throughput, and cost-effective analysis, with exceptional data quality and minimal sample.
Email: marketing@mtoz-biolabs.com
