

- Home
- Companies
- Applications
- Software Solution for Remote Crop ...
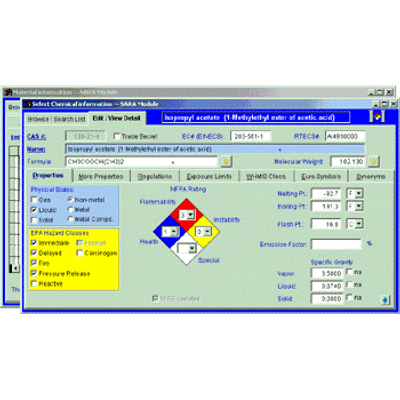
SARA - Tier Reporting & Inventory Management - SARA Module
To manage your materials and chemical components data, and to comply with your Federal, state and local Tier reporting requirements, consider the following MIRS solutions: SARA module, LOCAL module, CLE module, INVENTORY module.
This standalone module provides Title III, Section 302, 304, 311 & 312 compliance with Tier reports, emergency release (spill) notification, LEPC notification and SDS chemical list compliance reporting.
- Generates Federal or State Tier II magnetic media for EPA submission.
- Includes the SARA Title III chemical database.
- Combines with the CHEMICAL LIST EXTENSION (CLE) for extensive agency cross reference.
An easy to useSARA Wizardsteps you through data entry and reporting for compliance with State or Federal Tier II requirements. If an accident involving a potentially hazardous substance occurs, use theEmergency Release Notification Wizardto determine if any reportable quantities were exceeded and, if necessary, to prepare a notification letter for local, state, and Federal agencies. Because the material tracking information is shared with other modules, such as FORM R and AIR, data entry burden is reduced, while the accuracy of material information is increased.
LOCAL module
This add-on to the SARA module provides State and Local Tier reporting addressing reporting variations from the EPA.
Specific implementations are available as well as Generic, a user-defined template for local reporting. Choice of specific implementations include: California, Delaware, Florida, Georgia, Kansas, Louisiana, Michigan CMR, Washtenaw County (Michigan), Minnesota, New Jersey, Ohio, Texas, Wisconsin.
CLE module
The CLE (Chemical List Extension) is an add-on module to SARA, FORM R, MSDS, AIR, WASTE or NPDES. The CLE greatly expands the SARA chemical database for identifying and cross-checking multiple agency lists.
CLE provides an integrated 36,000+ chemical library to expedite entry of chemical ingredients, and cross reference for more than 50 agency lists, including SARA, CAA, TSCA, ODC, and various state lists, such as CA, MI, NJ, NY, PA, WI.
INVENTORY module
INVENTORY is an add-on module to SARA, FORM R, or MSDS. INVENTORY provides `real time` chemical inventory tracking by location or by container throughout the year.
- Tracks transactions by location for physical inventory, purchase, usage and inventory transfers.
- Calculates annual amounts from the transactions and integrates with the SARA and FORM R modules for SARA Title III reporting.
The screen below is from the Material Tracking database. The Material Tracking database contains the information needed for Federal or State Tier II and EPA SARA Title III Section 313 (TRI) reporting. Here you can track information about the materials used in your facility, the storage location and amounts.
Much of this Material Tracking information is shared with other compliance tasks, such as creating a Waste Manifest or reporting emissions under Title V of CAA. Using MIRS™, with its integrated modular system, makes all your compliance tasks easier.
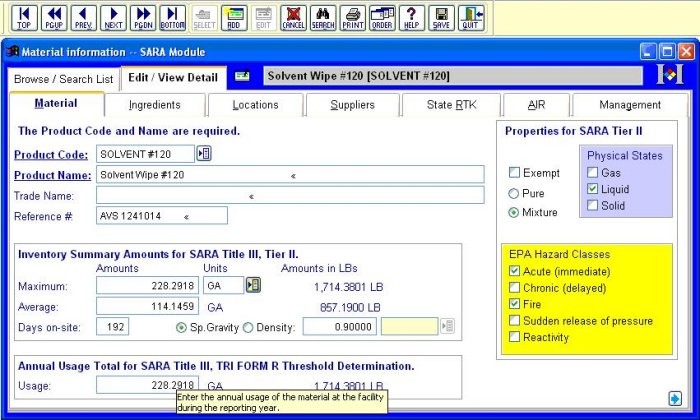
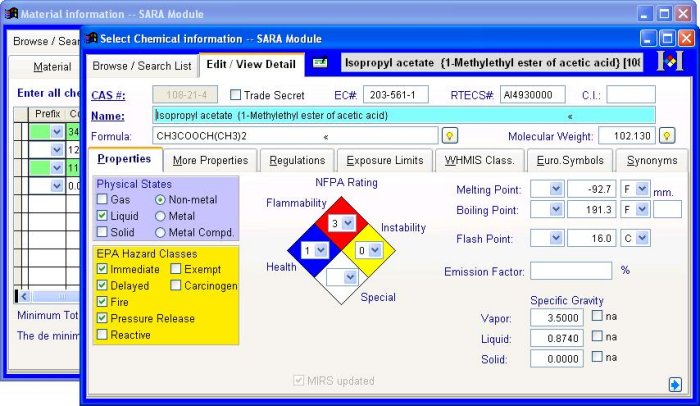
All MIRS™ major modules include the Regulated Chemical database,a complete EPA-regulated database of approximately 1800 chemicals. This database covers all chemicals regulated by SARA Section 302 (Extremely Hazardous Substances list), Section 304 (Comprehensive Environmental Response, Compensation and Emergency Liability Act), and Section 313 (Toxic Release Inventory). The Regulated Chemical database saves time, as you simply select the material`s chemical components from the pull down list.
For chemicals found in the Regulated Chemical database or the add-on Chemical List Extension (which brings the number of chemicals in the database to more than 37,000) related chemical information fills in automatically. This chemical information can include regulatory status, physical states, hazard classes and more.
The printed report is acceptable for direct submission to applicable state or local agencies that accept hard copies. The electronic version of the report in the EPA`s Tier 2 Submit format is also available and is acceptable for submission.

