- Home
- Companies
- Magritek Ltd.
- Applications
- Benchtop NMR spectrometer solutions for ...

Benchtop NMR spectrometer solutions for analytical NMR sector - Monitoring and Testing - Laboratory Equipment
NMR is an excellent tool for quantification and identification. The peaks in the spectrum will appear in predictable locations in the spectrum and their intensities are proportional to the number of nuclei in the sample.
NMR is best known for its use in structure determination. The chemical structure of a small molecule dictates a rational spectrum. This is because the electronic structure, the bedrock of chemistry, is what determines the chemical shift, while the number of nuclei determine the coupling and relative intensities.
These same properties result in NMR being excellent for quantification and identification. The peaks in the spectrum will appear in predictable locations in the spectrum and their intensities will be proportional to the number of nuclei in the sample. In other words, the peaks are a chemical signature and the sample concentration is proportional to the intensity of the NMR signal detected. Sample concentrations and purities can be easily measured from known peaks once the proportionality constant is calibrated using a reference of known concentration. Using these peak positions and relative intensities, the compounds, mixtures and impurities in a sample can be identified or quantified.
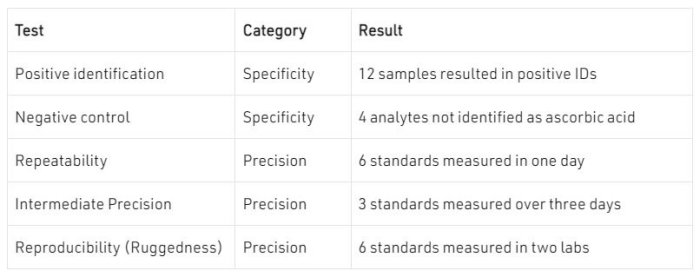
NMR spectra can be used to identify compounds. Depending on the degree of specificity required, this can be a simple peaks comparison or can incorporate other tests related to peak splitting and integral intensities. Here we show a method for identification of ascorbic acid, which is a common vitamin sold on its own as well as in many dietary supplements.
The United States Pharmacopeia method for identification of ascorbic acid requires both FT-IR and a reducing sugar chemical test because neither has the specificity on its own. Here a single NMR spectrum is acquired and analysed with three different tests: peaks, multiplet, and nuclides count comparisons to quickly identify ascorbic acid. A similar set of tests is used for sodium ascorbate.
The peak positions in the spectrum are compared to a peaks list determined from a standard material. If the peaks match, then the sample passes the peaks comparison test. The peak positions are shown above the spectrum and range from 3.64 to 4.99 for this sample.