

- Home
- Companies
- Dispersion Technology, Inc. (DTI)
- Articles
- Adsorption of surfactant from non-polar ...
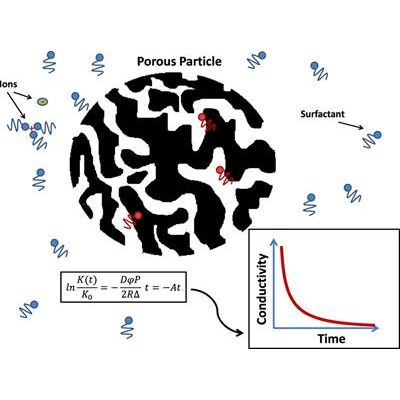
Adsorption of surfactant from non-polar liquid into porous material characterized using conductivity measurement
Hypothesis: Surfactants cause ionization in non-polar liquids, enabling such liquids to become electrically
conductive with that conductivity being a linear function of the surfactant concentration. Consequently,
measurement of the conductivity can be used as a tool for monitoring surfactant concentration.
Experiments: We describe here a simple method for studying surfactant adsorption from oil into a porous
material. The conductivity of solutions containing toluene and porous particles was measured as a
function of time after the addition of surfactant, at various concentrations. We applied this method for
characterizing surfactant (SPAN 20) adsorption by porous particles (silica gel Davisil) suspended in the
non-polar liquid (toluene). We the suggested a simple theoretical model for the initial stage of this
adsorption process and tested its prediction experimentally.
Findings: The experimental data confirms all predicted theoretical trends both qualitatively and quantitatively.
This method can be used for understanding surfactant behavior in rock formations during oil
recovery, optimizing surfactant concentration, and analyzing chemical composition.
