

- Home
- Companies
- Jenfitch INC.
- Articles
- Cooling Tower Water Treatment In ...
Cooling Tower Water Treatment In Southeast Texas - Case Study
For nearly a decade, a seasoned water treatment technician had managed a 60,000-gallon open counter-flow cooling tower at a manufacturing facility near Houston, Texas. The system faced a persistent challenge: a water pH of 8.5.
Traditional oxidants like sodium hypochlorite were ineffective under these conditions, forcing the team to rely on a complex chemical program:
- Solid Bromine tablets applied weekly
- Continuous ozonation requiring compressors and air dryers that demanded weekly maintenance
- High-strength hydrogen peroxide (35%), applied twice monthly (and weekly during summer)
- A dispersant and a corrosion inhibitor will continue to be applied weekly.
Fig 1 (left). View of the air intake and water basin. The slime shown was typical for the time of year. This photo was taken in September 2019.
Despite these efforts, the tower still suffered from:
- Algae overgrowth and biofilm accumulation
- Poor microbial control, even after high-dose peroxide shocks
- Operational headaches from maintaining ozone systems and handling hazardous chemicals
- No improvement when alternative chemistries, like peracetic acid, were tested
The treatment program was costly, chemical-intensive, labor-intensive, energy-intensive, and still not effective in the delivery of reliable microbial control.


Routine quality checks included bromine residual, pH, chlorides, total iron, hardness, silica, and conductivity, along with dip-slide sampling for bacterial growth. These samples frequently showed significant colonization. In fact, shock treatments with hydrogen peroxide often left visible regrowth within a couple of days.
Below, Fig 4 on the left shows a typical colonization with the Bromine/Ozone treatment program; Fig 5 on the right shows the incubated dip-slide results of a water sample taken after a 35% hydrogen peroxide shock application.

Turning Point: A Trial with Mineral Oxychloride Technology
In 2024, the facility temporarily shut down during a change of ownership. The cooling tower was turned off but not drained, leaving water stagnant in the basin for eight months. By the time operations resumed in March 2025, the technician was determined to adopt a better solution.
After conducting some research, he identified mineral oxychloride advanced oxidation technology as a potential game changer and decided to test it. Because the customer does not have dosing equipment, the chemical treatment will be applied as a slug dose every 7 to 10 days. His objectives were clear:
- Replace bromine and ozonation with a simpler, more effective oxidant
- Achieve consistent biofilm and algae control without the recurring complications of peroxide and ozone systems
- Reduce overall chemical, energy, and labor costs
March 26th. 2025
On the first day of the Mineral Oxychlorides (MOCI) protocol, conditions were severe: the stagnant basin water was heavily polluted with organic matter and further contaminated by an industrial effluent spill that was disposed of in the basin. Even under these circumstances, he moved forward with the new program.
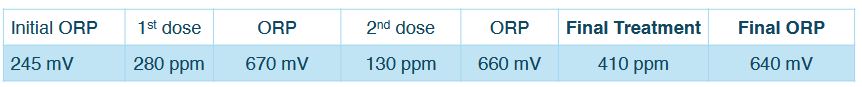
Notes: The treatment aimed to increase the initial ORP to a minimum target of>+600 mV.A measurement taken five minutes after the first 280 ppm dose of mineral oxychloride AOP showed the ORP had reached 670 mV.A second reading was taken 30 minutes later, after the oxidative energy in the system had stabilized. Because the ORP had dropped significantly, a second dose was administered to reach the target level. Five minutes later the ORP had increased to 660 mV. The technician returned about 20 minutes later to record the final stabilized reading of640mV.
MARCH 31st 2025
In the following weeks, as the system became progressively cleaner, the dosage of mineral oxychlorides was reduced with each visit. Only half of the dosed applied the previous week was necessary to stabilize the system at 644 mV. The free chlorine residual corresponding to the final ORP level was measured at 2 ppm.

April 14™. 2025
Just three weeks after the initial treatment, the MOCI dosage had already been reduced by 73%.
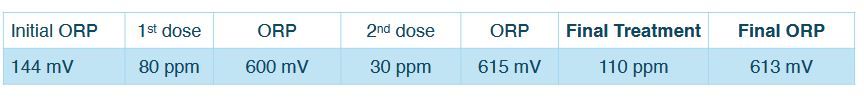
MAY 7™. 2025, MAY 15th. 2025, MAY 23™. 2025
After six weeks with the new MOCI protocol the system stabilized at a weekly dose of 80 ppm.

A significant amount of rainfall occurred during May, increasing the organic load of the local river that serves as the water source. The MOCI dosage was maintained at the same level, even though some variation in the final ORP was observed (550 mV to 620 mV) due to higher oxidative demand from the increased organic content coming in with the makeup water. Despite this, monitored water quality parameters and bacterial control remained stable.
Can Bleach Maintain Cleanliness and Stability in a System at a pH of 8.5?
The technician was very pleased with the results achieved in a relatively short period of time. The system required only a weekly visit for slug dosing and parameter monitoring, without the need to invest in new equipment for automatic ORP control or chemical dosing. He wondered whether, now that the biofilm had been eliminated from the system, maintaining sufficient oxidative energy with bleach might be adequate for a maintenance program.
For experimental purposes, during the month of June the mineral oxychloride reagent was replaced with sodium hypochlorite. A dose of 80 ppm MOCI had proven effective over the previous six weeks, and this dosage was used as baseline to dose 12.5% sodium hypochlorite. The same monitoring was conducted. Water quality parameters, including ORP readings and chlorine residual, were within range. Therefore, the decision was made to maintain 80 ppm with the bleach trial.
However, three weeks into the new protocol, dip-slide samples showed a sharp increase in bacterial growth, indicating a loss of microbial control. As a result, the trial was terminated prematurely, without completing the one month originally planned. Please see the data below. Note that based on the weekly ORP readings, there was no indication that biofilm was building up, and that bacterial colonization was not being controlled. Free chlorine residual readings, not shown here, were also within range.
What happened? At a pH of 8.5, chlorine is not effective. Although the ORP readings were within the expected range because they reflected the oxidative energy present in the water, most of that energy was associated with the hypochlorite ion, a weak oxidant, rather than with the more powerful hypochlorous acid.
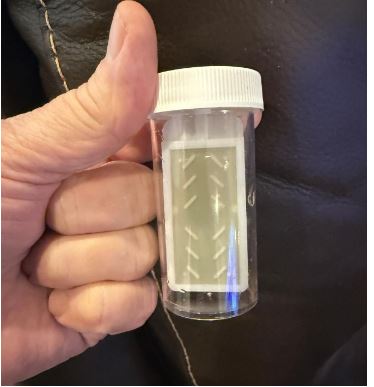
Fig 6. (Right) Photo of a dip-slide sample depicting the heavy bacterial colonization within 3 weeks of using sodium hvDochlorite as a stand-alone treatment.
June 1st, 2025 :

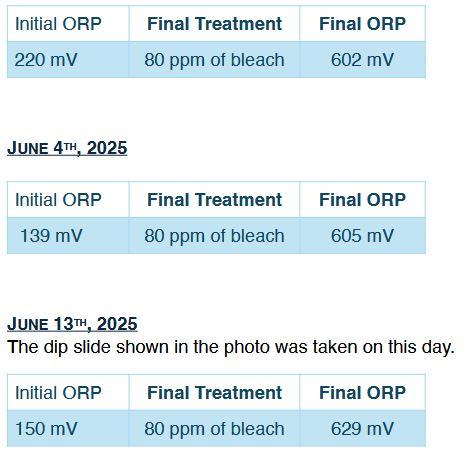
Can Mineral Oxychlorides and Bleach Work Together with a Synergistic Effect?
Mineral oxychlorides and bleach can be used together. The former can enhance the biocidal effectiveness, biofilm penetration, and residual activity of the latter. However, at a pH of 8.5, MOCI is doing all the work.
The technician wanted to try one more approach. Similar to how chlorine dioxide and bleach are sometimes used together to improve biocidal effectiveness and enhance biofilm control, he wondered whether replacing only a portion of the mineral oxychloride solution with bleach could reduce costs.
He initiated a second experiment using a combination of sodium hypochlorite and mineral oxychlorides, aiming to evaluate their complementary performance. The goal was to leverage the broad-spectrum advanced oxidation power of MOCI for bacterial and biofilm control, along with the lower cost of NaOCI.
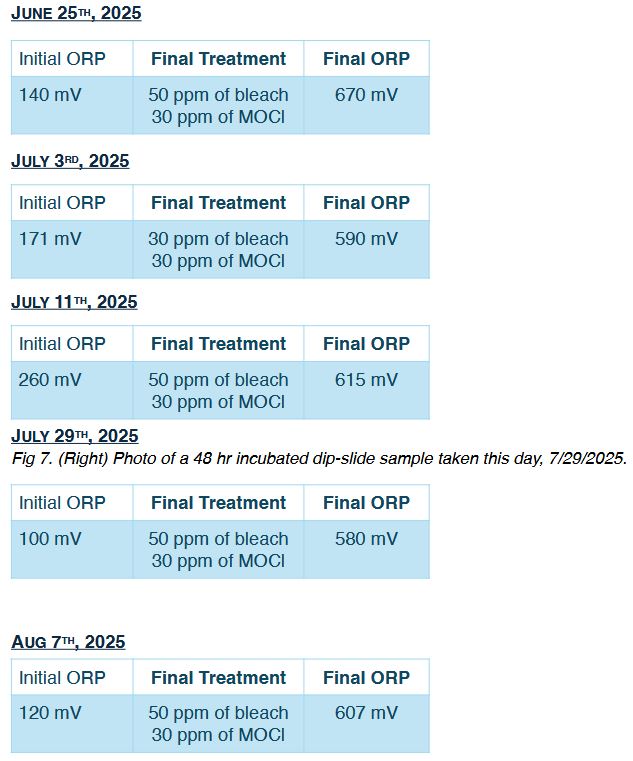
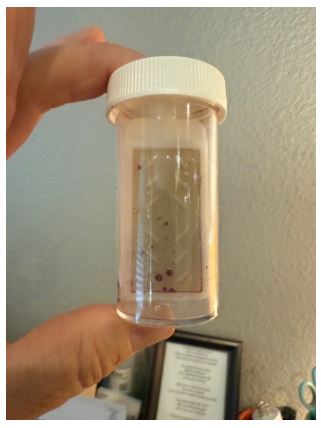
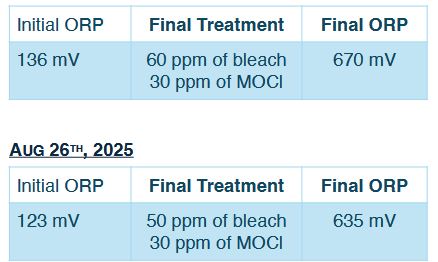
Figure 9 (left) shows 2019, before treatment
Figure 10 (right) shows following the oxychloride
The cooling tower basin in August with mineral oxychlorides.
The same basin in August 2025, implementation of mineral technology.


Trust the Process
If 80 ppm of bleach was not effective at controlling bacterial contamination in June due to a water pH of 8.5, what are the chances that a lower amount of bleach would be effective at the same pH?
It took some convincing, but by September the technician agreed that sodium hypochlorite was likely contributing very little or nothing at all, and should be removed from the program. The treatment protocol then resumed with mineral oxychlorides as a standalone disinfectant, this time at a reduced weekly dosage of 30 ppm instead of the original 80 ppm.
The data spoke for itself. During the previous two months, the water had been treated with 30 ppm of mineral oxychlorides in combination with up to 60 ppm of bleach. All monitored parameters stayed within operational limits. The system was kept clean, without visible algae, and bacterial control stayed consistent. The data indicated that 30 ppm of mineral oxychloride alone was sufficient to meet the oxidative demand of the system and control biofilm, while any contribution of sodium hypochlorite to microbial inactivation or residual protection remained questionable.
The study ultimately came full circle. At the time of concluding this evaluation, the cooling tower system had been maintained successfully for seven months with a weekly dose of only 30 ppm of MOCI. On a few occasions, the technician was unable to perform the regular weekly visit, and the system would go two weeks without treatment. Even in those instances, the monitored parameters were acceptable.
Effectiveness of Mineral Oxychloride AOP Reagent in High pH Cooling Systems
Reactive oxygen species (ROS) are the primary oxidants in mineral oxychloride advanced oxidation technology. Reactive chlorine species (RCS) are also present, but the system`s performance is driven by ROS dominance rather than halogen chemistry alone.
So, what does this mean in cooling tower operation?
Faster reaction kinetics
ROS react at significantly higher rates than chlorine and bromine because they require lower activation energy to operate. In mineral oxychloride solutions, the electrolyte structure is catalytic and continuously generates ROS, delivering consistent oxidative power without external energy input.
Greater oxidative capability
ROS have a higher oxidation potential than halogens. In practice, this translates into more effective degradation of resistant organic matter and deeper disruption of biofilm and cellular structures that conventional oxidants struggle to control.
Broader oxidative activity
ROS are inherently unstable, highly reactive, and non-selective. Rather than a single biochemical pathway, they damage multiple cellular components simultaneously, delivering stronger biological control and preventing microbial adaptation.
Improved diffusion into fouling layers
Due to their small size and higher diffusion rates, ROS interact more effectively with biofilms and microbial colonies. Halogens are often consumed before reaching deeper, protected zones.
All of this becomes even more relevant in high-pH cooling systems.
Chlorine, bromine, and to a lesser degree chlorine dioxide, lose effectiveness as pH rises. Mineral oxychlorides are less pH-dependent because ROS oxidation does not rely on acid-base equilibria. This makes MOCI technology a well-suited alternative for high-pH water systems currently running stabilized bromine or chlorine dioxide programs.
In field use, we consistently observe chlorine replacement ratios between 1:8 and 1:10; and bromine replacement ratios between 1:3 and 1:5. In practical terms, after stabilization, it typically takes eight to ten times less mineral oxychloride reagent to replace bleach, and three to five times less to replace activated liquid bromine in high pH cooling systems.
Switching to mineral oxychlorides commonly delivers both cost and performance advantages:
- Improved heat-transfer efficiency. Stronger biofilm control results in cleaner heat-transfer surfaces and better thermal performance.
- More effective slug dosing. Suppresses biofilm regrowth and maintains oxidative activity for longer intervals between treatments.
- Eliminate need for secondary biocides. Offers strong potential to eliminate or significantly reduce non-oxidizing biocidal alternatives.
- Lower product degradation. Compared with bleach, mineral oxychlorides degrade more slowly, which can extend delivery intervals from monthly to quarterly or longer.
- Simplified treatment programs. Streamlined feed strategies and safer handling procedures.
- Seamless implementation. Conversion typically requires no equipment changes, no process redesign, and no operational disruption. The only change is the oxidant feed source.
The mineral oxychloride reagent is EPA registered under FIFRA as a primary disinfectant and certified under NSF/ANSI 60 for use in potable water treatment applications.
