

- Home
- Companies
- Dispersion Technology, Inc. (DTI)
- Articles
- Ion-Pair Conductivity Theory V: ...
Ion-Pair Conductivity Theory V: Critical Ion Size and Range of Ion-Pair Existence
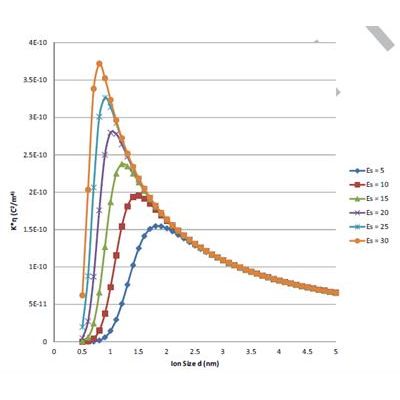
There are two mechanisms by which ion size affects conductivity of liquids: hydrodynamic resistance, and electrostatic attraction between cation and anion that leads to the formation of ion-pairs. Increasing ion size reduces conductivity due to growing hydrodynamic resistance. On the other hand, increasing ion size reduces the probability of ion-pairs formation, which leads to higher ionic strength and higher conductivity. The opposing nature of these two mechanisms indicates the existence of a “critical ion size” that corresponds to the maximum conductivity. We apply a theory that was published recently in this Journal with a wide variety of
supporting experimental data for determining value of this “critical ion size”. This theory predicts a very peculiar phenomenon: ions with sizes that are narrowly distributed around 1 nm (±0.5 nm) must dominate conductivity. This prediction is valid for solutions with ion-pairs, which according to the theory could occur when relative permittivity is below 10 if ionic strength of the liquids is low (<10−6 mol/l). This threshold shifts toward higher relative permittivities with increasing ionic strength.
