

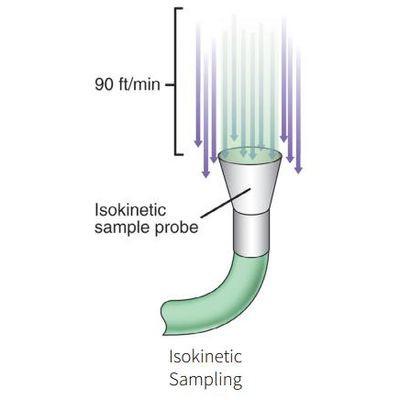
Isokinetic Sampling in Unidirectional Flow
Introduction
To maintain compliance with regulations and produce products with limited risk, pharmaceutical companies manufacture products in controlled environments such as cleanrooms. Cleanrooms reduce the risk of variability and particle contamination and are defined as rooms in which air filtration, air distribution, utilities, materials of construction, and equipment are maintained in a controlled manner.
One common contamination management technique that cleanrooms employ is unidirectional airflow. A cleanroom with unidirectional airflow utilizes a controlled, steady velocity of air in parallel streamlines that move in one direction to minimize the potential movement of contamination. Unidirectional cleanrooms are typically designed using laminar airflow hoods that direct the air, and the airflow is designed to wash particles from a potential contamination source away from the process risk before being exhausted from the process area. The constant laminar flow of clean air ensures that this potential contamination does not extend beyond a field of control.
