- Home
- Companies
- BMT USA, LLC
- Products
- BMT Steripro - cGMP Saturated Steam ...

BMT Steripro - cGMP Saturated Steam Sterilizers
Steripro saturated steam sterilizers are designed for production in pharmaceutical and biotech facilities where repeatability is critical. Equipped with high-grade vacuum pumps, non-proprietary components, and industrial-grade PLC control systems, the Steripro produces repeatable cycles with even temperature distribution better than +/- 0.5°C. Steripro sterilizers can be configured with custom chamber sizes, mechanical layouts, and process cycles to meet your specific requirements.
Chamber Construction
Highly polished 316L stainless steel chambers provide superior durability. The chamber door is also constructed with solid plate 316L stainless steel and held in place on all four sides by precision machined brackets for smooth travel and a tight seal against the chamber during the cycles. All chamber connections are sanitary clamp type and sloped to drain. Lubricant free door gaskets provide greater longevity and protect against particle shedding into clean rooms.
Sanitary Piping
All piping and ports in contact with the chamber are constructed of 316L stainless steel and sloped to drain with no dead legs. Sanitary clamp fittings are used for ease in removal of components. Non-proprietary sanitary valves and components are selected for their reliability and availability to reduce machine downtime.
Validation
Validation is a critical concern when purchasing a cGMP sterilizer, so special attention has been given to the documentation and repeatability of each system. Every BMT USA sterilizer is subjected to rigorous pre-validation testing at our facility and shipped with an extensive documentation package to help minimize the time required for onsite validation. IQ/OQ protocols and on-site execution options are also available.
Repeatability
The primary use of cGMP sterilizers is for production in pharmaceutical and biotech facilities where repeatability is critical. All BMT USA cGMP sterilizers utilize high-grade vacuum pumps, the highest quality instrumentation, and industry-standard PLC control systems to ensure repeatable cycles regardless of changing environmental conditions. Temperature distribution within the chamber is better than +/- 0.5°C
Clean Steam Source
BMT USA steam generators are cGMP-compliant and produce pure or clean steam for pharmaceutical sterilization. The generators can either be configured as stand-alone units for use as a central steam source or directly integrated into the sterilizer. BMT USA steam generators are assembled with the highest quality non-proprietary components and are available in electric or plant steam heated models.
Vertically integrated electric steam generator
BMT USA is one of the only manufacturers offering vertically integrated electric clean and pure steam generators up to 400lbs/hr with a maximum output pressure of 80 PSIG. Our vertical integration offers significant space savings while maintaining tight steam pressure control.
Control System
Industrial-grade Allen Bradley or Siemens PLC control systems provide superior operational reliability and cycle repeatability. The control systems are developed under the latest GAMP (Good Automated Manufacturing Practices) guidelines to ensure system reliability and ease in validation. The control system is also supported with a comprehensive documentation package.
The control system can configure and store 20 sterilizing cycles, which are accessed via a large touch screen operator interface terminal. In double door units, operator interface terminals are mounted on both the load and unload side of the sterilizer. All sterilizer functions are accessed through these screens and are secured by multi-level authentication. If electronic data storage is desired, the control system can be provided with 21 CFR part 11 compliance. Printers and recorders can also be equipped for physical data storage on paper.
Custom Loading Equipment
BMT USA manufactures stainless steel loading equipment in standard or custom designs to accommodate loads of any size, shape or weight. Our smaller chamber sizes can be equipped with wire or perforated shelves and loading racks, while larger chambers can be equipped with carts and transfer trolleys.
Large pit-mounted chambers include options for tracks and rub rails that can be designed to work with the wheel configuration of your existing equipment. Carts with drop down wheels are available for rooms with limited space.
Sanitary Stainless
The chamber, chamber door, sanitary piping and fittings are all constructed of 100% 316L stainless steel.
Non-Proprietary
Fully constructed with high-grade non-proprietary components for improved reliability, serviceability and value.
Temperature Uniformity
Temperature distribution inside the chamber better than ± 0.5°C
Compliance
Fully engineered and built in compliance with US FDA current Good Manufacturing Practice (cGMP), Good Automated Manufacturing Practice (GAMP), and US FDA CFR 21 Part 11.
Energy Efficient
The chamber and all piping systems are fully insulated to help maintain temperatures and reduce utility consumption.
PLC Controls
Industrial-grade Allen-Bradley or Siemens PLC-based control systems deliver reliable performance and repeatable operation.
Automatic Doors
Automatic pneumatic driven precision sliding chamber doors improve safety and convenience during operation.
Lubricant-Free Door Gasket
Lubricant-free door gaskets provide greater longevity and eliminate particle shedding into clean rooms.
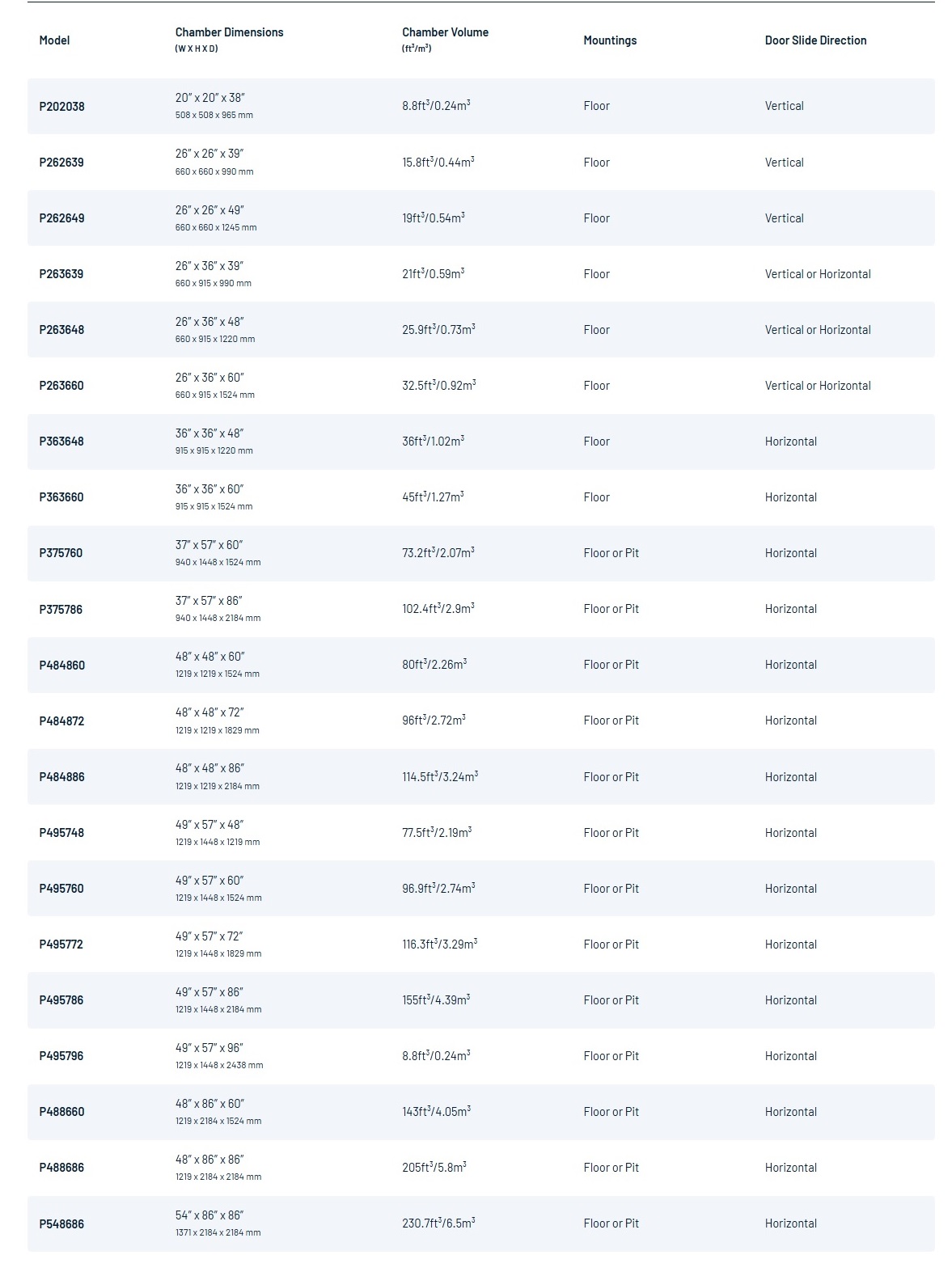
- Custom chamber size and capacity.
- Right or left service area access.
- Floor or pit mounting.
- Integrated or remote controls.
- Single-door or 2-door pass-through.
- Custom sterilization cycle recipes.
- Custom loading equipment, including carts, trolleys, shelving and trays.
- Integrated or standalone steam generator / electric or plant steam heated.
Effective sterilization of hard goods, filters, linens, porous materials, wrapped goods, and products that are unaffected by vacuum.
The load is preconditioned with alternating vacuum and steam pulses, then heated to an exposure temperature between 110°C and 135°C. The exposure temperature is maintained for a specified period, after which rapid drying begins with fast exhaust and pulling a deep vacuum.
Dry Goods Cycle - Pre-Vacuum with Air Pulse DryingEffective sterilization of rubber stoppers, hoses, and other material where trapped moisture can be difficult to remove.
The load is preconditioned with alternating vacuum and steam pulses, then heated to an exposure temperature between 110°C and 135°C. The exposure temperature is maintained for a specified period, after which drying begins by fast exhaust, vacuum and air pulses which are highly effective in removing moisture from the load. The air pulses can also be heated to further aid the drying process.
Solution Cycle - Gravity Air Removal with Ramped ExhaustSterilization of liquids in vented or sealed glass containers
The load is preconditioned with steam pulses before heating to an exposure temperature between 110°C up to 135°C. The exposure temperature is held for a specified time period followed by a gradual exhaust ramping to cool the load back to ambient temperature.
Solution Cycle - Air Over-Pressure with Jacket Water CoolingSterilization of liquids in heat or pressure-sensitive containers
The load is preconditioned with steam pulses, then heated to an exposure temperature between 110°C and 135°C. The exposure temperature is maintained for a specified period, after which the cooling phase begins by injecting air at a pressure equal to that inside the product container to prevent damage or deformation. To accelerate cooling, cold water is introduced into the chamber jacket.
Steripro sterilizers can be programmed with any combination of saturated steam and vacuum sterilization cycles, with configurable parameters for each cycle phase. The programmed cycle recipes are securely stored in the control system memory and can be effortlessly retrieved and initiated via the touchscreen operator interface. Unauthorized modifications are prevented through a multi-level authentication system, ensuring recipe integrity and operational compliance.