- Home
- Companies
- PID Analyzers, LLC
- Products
- PID Analyzers - Model 33W - Compact ...
PID Analyzers - Model 33W -Compact Arsenic Analyzers in Water
Model 33W- Arsenic in Water (no organics) Tap water, well water or bottled water that does not contain volatile organic compounds does not need a GC for removal of VOC’s from the AsH3 peak so a GC is not needes and this version is less expensive.
- HG/GC/PID
- Hydride Generator
- Peltier cooler for moisture removal
- Concentrator/thermal desorber
- Computer:
- Pentium PC
- Operating System: Windows
- Color Touch Screen Display 8”
- Keyboard/Mouse
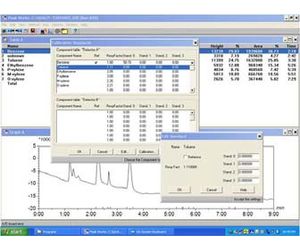
- PeakWorks-Chromayography/control software
- 8 Gbyte memory stick
- 16 bit ADC/ 4 ranges
- Autozero
- 5 timed evemts
- PID with 10.6 eV lamp
- NEMA 2 Enclosure- General purpose
- 19" W x 9"H x 16"D;
- Weight- 18 pounds
- Standard output: 0-1 VDC, RS232; internet connection
Consumer Reports (January 2012) tested 5 brands of juices and found that 10% had total arsenic levels that exceeded federal drinking-water standards of 10 ppb. Dr. Brian Jackson & co-workers at Dartmouth College tested products sweetened with organic brown rice syrup, including cereal bars and baby formulas, and found ppb levels of arsenic that exceeded standards for for bottled water.
The FDA tests for total As. If the total As is > 23 ppb, a test for inorganic As is run. The standard is based on the concentrations of As(III) + As (V). The inorganic As compounds are more toxic than the organic forms monomethyl and dimethyl arsenic. The FDA standard for As in juices may be lowered in the near future.
A hydride is a compound in which one or more hydrogen atoms have reducing, or basic properties. In hydrides, hydrogen is bonded to a more electropositive element such as a metal. When arsenic compounds in solution are treated with a reducing agent (NaBH4), a hydride, is formed (AsH3,) and this is one of the most common pretreatment methods for determining aqueous arsenic.
More than 25 years ago, Cutter et al (1988) developed a method for detecting ppb levels arsenic in seawater using hydride generation/GC/Photoionization (PID). We have modified that PID method for As in drinking water and Foods.
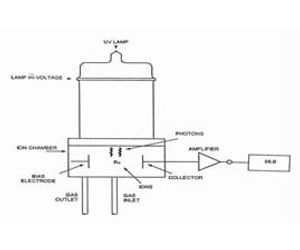
The PID uses a 10.6 eV lamp (the Ionization Potential for AsH3 is 10.6 eV )to ionize arsine (g) generated by reduction with NaBH4. When AsH3 absorbs a photon, it forms a positive ion AsH3 + plus an electron (e-). The positive ions are collected in an ionization chamber where the bias electrode pushes the ions to the collection electrode and the current collected is proportional to the concentration over a range of 107 from ppb to high ppm. A schematic of the PID is shown below.
The PID has been employed by the US EPA to develop more than a dozen standard methods for the measurement of ppb levels of VOC’s in drinking water.
If there are high ppb levels of organics in the water then the HG/ GC/PID method should be used to separate the AsH3 after the preconcentration phase to prevent any interference.
The organic level can be determined by simply running a blank without adding any NaBH4 to the sample.
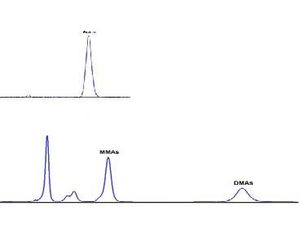
Inorganic Arsenic exists in nature in two forms As(III) and As (V) with the former being more toxic. Organic arsenic compounds such as monomethyl arsenic (MMA) and dimethyl arsenic (DMA) are less toxic than the inorganic form and have been used as a herbicide. As (III) & As(V) can be separated using the pH of the hydride generating solution (see below) and the resulting AsH3 detected by GC/PID shown below at a low ppb level (Courtesy Greg Cutter, Old Dominion Univ.)
The MMA and DMA are easily separated by GC and detected by the PID.
Arsenobetane, an organoarsenic compound that is found in fish that is supposedly non toxic.
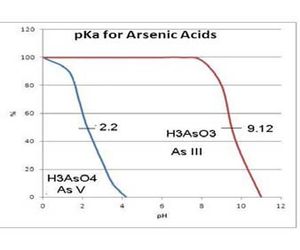
The inorganic arsenic compounds can be easily separated by pH as shown in the figure in the next column.
If the pH is in the 4-8 reglon (Tris buffer), the H3AsO3 is the predominant species while any As (V) exists as the anion and will not be converted to AsH3 by HG. By modifying the pre treatment both Cd and Pb can be detected with this analyzer.
Now if the pH is increased to
The MMA and DMA can be determined with the As (III), separated by a GC column and detected by a PID.
Hydride generation is a common pretreatment method for the measurement of aqueous As. For the PID method, it is different than the AA method. NaBH4 is added to a pH adjusted solution of As and the As is converted to AsH3 gas which is sparged from the solution and collected on a solid sorbent.
The analyzer is controlled by our Peak Works software that runs on Windows operating system on the embedded PC.