PMS - Model APSS-2000 -Liquid Particle Counter for USP 788
The APSS-2000 Liquid Particle Counter for USP 788 sizes and counts suspended particulate matter in a wide range of liquids, including parenterals, to meet all current U.S., European and Japanese Pharmacopoeia standards. Complete system includes: SLS-1000 Syringe Sampler.
The APSS-2000 is engineered to safeguard product integrity and patient safety by delivering uncompromising accuracy and compliance in liquid particle monitoring.
Data Integrity & Compliance
- 21 CFR Part 11 Ready: Electronic records and signatures supported through SamplerSight Pharma software.
- Validation Built-In: IQ, OQ, and PQ protocols included for full system qualification.
- Secure Architecture: Active Directory integration and automated backup scheduling protect your data.
Operational Efficiency
- Recipe Functions: Standardize workflows to minimize operator error and ensure consistent compliance.
- Automated Calibration: Full calibration or routine verification at the touch of a button.
- Custom Reporting: Flexible formats for streamlined audits and quality reviews.
Precision Sampling:
- 100% Sample Volume Accuracy: Every drop counts—ensuring complete measurement for reliable results.
- Syringe-based control achieves repeatable volumes within 0.4 mL, reducing variability and risk.
Complete Reporting
- SamplerSight Pharma Software allows operators to manage sampling requirements for batch-based operations and provides a comprehensive view of the batch information with histogram, time plot and tabular data presented in an easy-to-use format that is easily reported. SamplerSight Pharma features a validatable, user-friendly, Windows-based software with context-sensitive help.
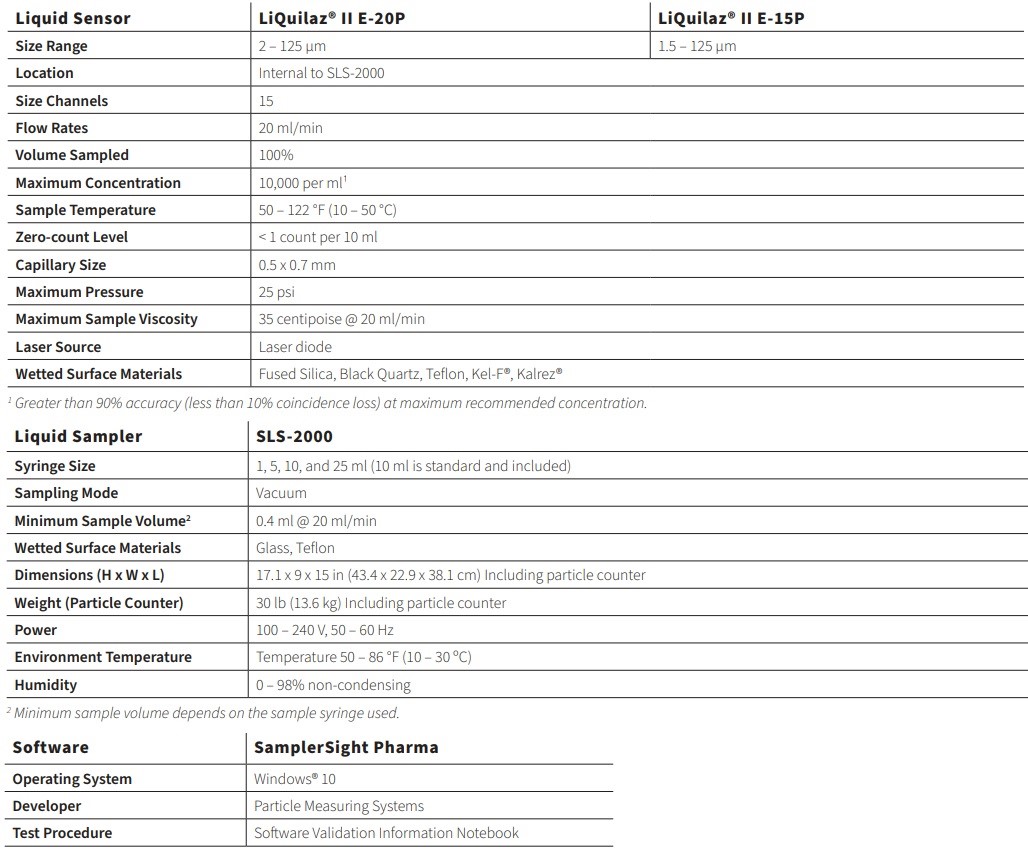
The APSS-2000 syringe sampling system sizes and counts suspended particulate matter in a wide range of liquids and meets or exceeds all current USP, EP, and JP requirements. Recipe functions allow the APSS-2000 to adapt to future regulatory changes.
The APSS-2000 system consists of an SLS-2000 syringe sampler, a LiQuilaz® II E Series light obscuration spectrometer, and SamplerSight Pharma software, allowing reports to meet 21 CFR Part 11 regulations.
Defect Reduction
- 100% sample volume for greater accuracy
- A precise sample volume within 0.4 ml is achieved with the syringe sampler, producing repeatable results
- SamplerSight Pharma software comes with a Validation Information Notebook, providing comprehensive software development
- documentation and full IQ, OQ, and PQ protocols
- SamplerSight Pharma complies with FDA 21 CFR 11
User-Friendly
- Reduce operator error and ensure process accuracy with recipes for repeat sample processing to USP, EP, or JP standards
- Automated particle counter calibration wizard for full calibration or routine verification
- Alarm levels for pass/fail criteria ensure quality control
- Automated backup scheduling ensures enhanced data integrity
Comprehensive System
- Easy-to-use pull-down menus
- Active Directory security password maintains system integrity
- Particle measurements are reported per ml or per container values
- Small sample volume minimizes waste of expensive product
- Various custom report formats are available
- Electronic report approval
- Particle size capability from 1.5 to 125 µm
- Fifteen user-selectable sizing channels
- Meets and exceeds all USP, EP and JP requirements
- Sample volumes from 0.4 ml to 1 liter
- Adapters for SVI and IV bag testing
- Automated sensor calibration
- Plug and play ultrasonic bath and magnetic stirrer options available
- Pharmaceutical parenteral monitoring to U.S., EP, JP and FDA standards
- Parts/medical device cleanliness testing
- Laboratory water sampling for purified water testing and water for injection (WFI)
- Filter efficiency testing