

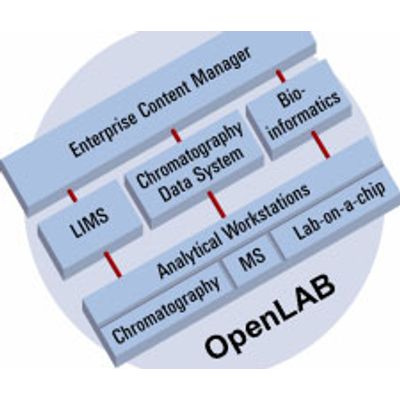
OpenLAB
OpenLAB is the latest innovation from Agilent Technologies for the laboratory. A scalable, feature-rich operating system for instruments and laboratory information, OpenLAB will provide you with a strategy for integrating instrumentation, local data systems, and laboratory electronic information into a fully protected, searchable, and archivable system. Utilizing the latest advances in software and database design, OpenLAB becomes your way of addressing present and future instrumentation needs, protecting and sharing your laboratory information safely and securely, and integrating and managing total laboratory information content.- Instrument Control Manager (ICM) provides a single system interface for controlling your entire laboratory
- Control, acquisition and processing of data from more than 300 instrument modules from 25 different instrument manufacturers within a modern web-based application.
- Data viewers to view chromatographic or mass spectral data without installing native applications or using proprietary file conversion.
- Enterprise Content Manager (ECM) provides controlled access to content and applications via permissions set by the system administrator
- Establish policies and manage file lifecycles automatically using archiving and record retention features
- Seamless integration of other CDS and workstations like EZChrom, ChemStation and Cerity for pharmaceutical QA/QC.
- Access any electronic file, anytime, anywhere.
- Capability to mine lab information through versatile database queries.
- Optional Business Process Manager (BPM) for workflow automation.
OpenLAB takes advantage of a vast library of control software that has been developed for more than 300 different instruments from leading chromatography manufacturers. Instruments ranging from Agilent 6890 GCs, 1100 HPLCs, Waters Alliance HPLC, Thermo HPLC and GCs and chromatography devices ranging from Alltech and ESA detectors, Alcott Autosamplers, and PE Nelson Intelligent Interfaces can all be added to any OpenLAB framework. OpenLAB`s extensive instrument control software enables laboratories that incorporate a multi-vendor, heterogeneous mixture of instruments to integrate everything under a single software platform.
Features
- Instrument control software and chromatography data analysis of more than 300 different HPLC, GC, and other chromatography systems from more than 25 different leading manufacturers. Complete Instrument Control List
- Networked data acquisition of all instruments through the Agilent Instrument Controller networked appliance.
- Client access to all instruments through web clients to enable real time access and control of instruments.
- Application of Enterprise Content Management (ECM) to non-chromatographic instruments (as well as electronic information from other software applications).
- Content management for instruments including full searching and sorting of information, on-line archiving, integration of instruments and maintenance operations, and more.
- When coupled with the Business Process Manager (BPM), instrument maintenance schedules can be automated and instruments can be locked down from use when maintenance is not performed in a timely manner.
OpenLAB Enterprise Content Manager (ECM) is a software solution that helps you make better decisions faster than ever. By providing a secure, central repository and rich content services, ECM allows you to create, manage, collaborate, archive, and re-use all of your business critical information with ease. ECM manages raw data and human readable documents of any data, in any form, from any supplier, and its web-based user interface drastically reduces the learning curve for new users.
OpenLAB ECM is a proven solution that has helped organizations in many industries get control of their business critical information while improving regulatory compliance, reducing operational costs and boosting productivity levels.
Features
- Manages raw data and human readable documents of any file type
- Meta data extraction from analytical and all Word processing applications. Complete list of Automated Data Extraction Services
- Central Storage and easy integration of MS Office data within the secure OpenLAB ECM data repository
- Full scalability from workgroup implementation to enterprise wide deployment
- Compliance with all major regulatory guidelines including 21CFR part 11
- Integrated Business Process Management module for automating and securing mission critical business processes
- Integrated archival and record retention policy modules
- Design for ease-of-use
In today`s business environment, organizations are faced with addressing numerous challenges, ranging from improving employee productivity, reducing operational costs, reducing business risk arising from non-compliance with corporate and regulatory mandates (e.g. Sarbanes-Oxley, 21 CFR Part 11, HIPAA), and accurately tracking key operational and performance metrics. Conquering these challenges amidst mounting competitive pressures, limited resources, numerous mergers and acquisitions, and exponentially growing and changing business processes can be a daunting task.
Features
- Streamline, automate, and optimize mission-critical business processes while enabling seamless collaboration between people, processes and content
- The entire solution is designed with ease-of-use in mind, so there is zero coding required to define, configure, and deploy a business process
- User interfaces are based on familiar productivity tools that every knowledge worker understands, such as Microsoft Internet Explorer, Visio, Outlook, Excel, and Adobe Designer (for eForms).
- Pre-screen analytical results stored in the Enterprise Content Manager (ECM) with the `metadata rule` process activity
- Manage instrument maintenance schedules when used with the Instrument Control Manager (ICM)
- Integration with Adobe Designer and OpenLAB for managing forms-based processes
- Optional software for OpenLAB
