Particle Measuring Systems (PMS) articles
GMP cleanroom contamination monitoring is a critical safeguard, forming the frontline defense against risks that can compromise product quality. When executed with intention and rigor, it becomes one of your most powerful tools for detecting contamination risks early and stopping issues before they escalate.
Aligned with EU GMP Annex 1, these ten practical tips are designed to help you build a monitoring program that is not only robust and defensible
Need a fast way to check cleanroom contamination limits?
Get your free quick-reference guide here.
That’s why Particle Measuring Systems has created the Cleanroom Standards At-a-Glance Cards, a free quick-reference tool designed to help you navigate cleanroom standards with ea
Introduction
To maintain compliance with regulations and produce products with limited risk, pharmaceutical companies manufacture products in controlled environments such as cleanrooms. Cleanrooms reduce the risk of variability and particle contamination and are defined as rooms in which air filtration, air distribution, utilities, materials of construction, and equipment are maintained in a controlled manner.
One common contamination management technique that cl
Introduction
The Technical Committee (ISO/TC209) responsible for ISO 14644, the set of Standards governing cleanrooms and associated controlled environments, looked at the need for a new document to aid users in the application of airborne particle counters; especially considering the new revision to EU Annex 1 – Manufacture of Sterile Medicinal Products (2022). The Technical Report (TR) describes the application of particle counters for classification and monitori
Introduction
Utilities can be classified as process systems or process support systems based on their use. Utilities that do not affect product quality or patient safety in any way are normally called process support systems (e.g., HVAC). All utilities in direct or indirect contact with the product or with critical materials as well as utilities in contact with components such as piping, valves, or pumps are considered process systems and are critical because they can po
Introduction
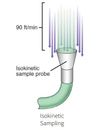
Microbiological sampling within a pharmaceutical manufacturing facility is performed using several methods:
- surface sampling
- passive aerosol (air) environmental sampling
- active aerosol (air) environmental sampling
Each of these methods produces a result based on the growth of microbial colonies on the surface of a collection media (typically agar), but the specific instruments and techniques vary between the
In controlled environments like cleanrooms, maintaining sterility is crucial. One of the most important processes in this context is microbial air sampling—a method used to detect airborne contaminants that could jeopardize sterile manufacturing processes. However, one significant challenge in microbial air sampling is the risk of false positives in cleanrooms. These false positives can lead to unnecessary investigations, downtime, and potenti
In the pharmaceutical manufacturing industry, ensuring sterility and minimizing microbial contamination are critical for maintaining product quality and safety. Microbial sampling is a key component of environmental monitoring, and understanding the optimal criteria for sampling can significantly impact the effectiveness of sterility assurance programs.
In the pharmaceutical industry, maintaining stringent contamination control strategies is not just a regulatory obligation but a fundamental aspect of ensuring product safety and efficacy. The EU GMP Annex 1 outlines the importance of a Contamination Control Strategy (CCS) for sterile drug products, emphasizing the need for a structured approach to manage risks and maintain compliance. We recently hosted a webinar
In the pharmaceutical industry, maintaining a controlled environment in cleanrooms is critical for product integrity and compliance. The recent CrowdStrike incident caused significant IT outages globally, affecting flight control, banking, logistics, and pharmaceuticals.
Our FacilityPro® environmental monitoring system showcased exceptional performance during this recent event, ensuring uninterrupted and continuous cleanroom monitoring and control. FacilityPro is a vital component